Abstract
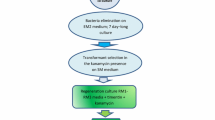
Hypocotyl, stem, leaf and shoot-apex of one-week-old seedlings ofVigna aconitifolia were inoculated with the nopaline wild type (T37) ofAgrobacterium tumefaciens to induce tumours. Hypocotyl segments of 1 cm length fromin vitro grown plants were also co-cultivated with a bacterial suspension to obtain transformed tissue. Axenic cultures obtained from both the experiments when cultured on basal medium proliferated into callus which differentiated into shoot buds on the same medium after, repeated subcultures. The transformed nature of the shoots was confirmed by their failure to produce roots, growth on hormone-free medium and the presence of nopaline and nopaline dehydrogenase.
Similar content being viewed by others
References
Akiyoshi D E, Morris R O, Hinz R, Mischke D S, Kosuge T, Garfinkel D J, Gordon M P and Nester E W 1983 Cytokinin/auxin balance in crown gall tumours is regulated by specific loci in the T-DNA;Proc. Natl. Acad. Sci. USA 80 407–411
An G, Watson B D, Stachel S, Gordon M P, and Nester E W 1985 New cloning vehicles for transformation of higher plants;EMBO J. 4 277–284
Deak M, Kiss G B, Koncz C and Dudits D 1986 Transformation ofMedicago byAgrobacterium mediated gene transfer;Plant Cell Rep 5 97–100
Eapen S, Gill R and Rao P S 1986 Tissue culture studies in mothbean;Curr. Sci. 55 707–709
Eapen S, Köhler F, Gerdemann M and Schieder O 1987 Cultivar dependance of transformation rates in mothbean after co-cultivation withAgrobacterium tumefaciens;Theor. Appl. Genet. 75 207–210
Garcia J A, Hille J and Goldbach R 1986 Transformation of cowpeaVigna unguiculata cells with an antibiotic resistance gene using a Ti-plasmid-derived vector;Plant Sci. 44 37–46
Gill R and Eapen S 1986 Plant regeneration from hypocotyl protoplasts of mothbean (Vigna aconitifolia);Curr. Sci. 55 100–102
Horsch R S, Fry J E, Hoffmann N L, Eichholtz D, Rogers S G and Fraley R T 1985 A simple and general method for transferring genes into plants;Science 227 1229–1231
Köhler F, Golz C, Eapen S, Kohn H and Schieder O 1987 Stable transformation of mothbeanVigna aconitifolia via direct gene transfer;Plant Cell Rep. 6 313–317
Lin M and Staba J 1961 Peppermint and spearmint tissue cultures. I callus formation and submerged cultures;Lloydia 24 139–145
Lippincott J A and Lippincott B B 1967 Time required for tumour initiation byAgrobacterium tumefaciens on pinto bean leaves;Nature (London)213 596–598
Mariotti D, Davey M R, Draper J, Freeman J P and Cocking E C 1984 Crown gall tumorigenesis in the forage legumeMedicago sativa L.;Plant Cell Physiol. 25 473–482
McCormick S, Niedermeyer J, Fry J, Barnason A, Horsch R and Fraley R 1986 Leaf disc transformation of cultivated tomato (L. esculentum) usingAgrobacterium tumefaciens;Plant Cell Rep. 5 81–84
Murashige T and Skoog G 1962 A revised medium for rapid growth and bioassays with tobacco tissue cultures;Physiol. Plant. 15 471–497
Nester E W, Gordon M P, Amasino R M and Yanofsky M G 1984 Crown gall: A molecular and physiological analysis;Annu. Rev. Plant Physiol. 35 387–413
Otten L A B M and Schilperoort R A 1978 A rapid microscale method for the detection of lysopine and nopaline dehydrogenase activities;Biochim. Biophys. Acta 527 497–500
Owens L D and Cress D E 1985 Genotypic variability of soybean response toAgrobacterium strain harboring the ti or Ri plasmids;Plant Physiol. 77 87–94
Petit A, Stougaard J, Kuhle A, Mareker K A and Tempe J 1987 Transformation and regeneration of legumeLotus corniculatus: A system for molecular studies of symbiotic nitrogen fixation;Mol. Gen. Genet. 207 245–250
Shahin E A, Spielmann A, Sukhapinda K, Simpson R B and Yashar M 1986 Transformation of cultivated Alfalfa using disarmedAgrobacterium tumefaciens;Crop Sci. 26 1235–1239
Schroder G, Waffenschmidt S, Weiler E W and Schroder J 1983 The T region Ti-plasmid codes for an enzyme synthesising indole-acetic acid;Eur. J. Biochem. 138 387–391
Vlachova M, Metz B A, Schell J and de Bruijn F J 1987 The tropical legumeSesbania rostrata: Tissue culture, plant regeneration and infection withAgrobacterium tumefaciens andrhizogenes strains;Plant Sci. 50 213–223
Webb K J 1986 Transformation of forage legumes usingAgrobacterium tumefaciens;Theor. Appl. Genet. 72 53–58
White D M R and Greenwood D 1987 Transformation of forage legumeTrifolium repens L. using binary Agrobacterium vectors;Plant Mol. Biol. 8 461–469
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gill, R., Eapen, S. & Rao, P.S. Transformation of the grain legumeVigna aconitifolia Jacq Marechal byAgrobacterium tumefaciens, regeneration of shoots. Proc. Indian Acad. Sci. 98, 495–500 (1988). https://doi.org/10.1007/BF03053407
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF03053407