Summary
-
1.
The polarographic behaviour of cobalt has been studied in ethanolamines employing the following electrolytes to alter the pH of the solutions: (i) Sodium acetate-acetic acid, (ii) Sodium carbonate, (iii) Sodium hydroxide, and (iv) Ammonium chloride-Ammonium hydroxide.
-
2.
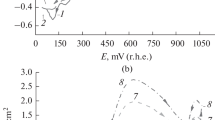
Studies of the effect of pH have indicated that in acidic solutions aquo-cobalt ion is reduced at the dropping mercury electrode. As the pH is increased to 9, the amine complex is formed, and when the pH is increased still further, hydroxyl group also enters into the complex.
-
3.
The reduction of the ethanolamine complexes of cobalt at the dropping mercury electrode is essentially an irreversible process except in solutions containing sodium carbonate and monoethanolamine.
-
4.
The foregoing studies indicate the formation of the following complexes of cobalt:—
$$\begin{gathered} (1) [Co (Moen)_1 (OH)_x ]^{2 - x} (2) Co (Moen)_2 (CO_3 )_1 \hfill \\ (3) Co (Dien)_2 (CO_3 )_1 (4) Co (Moen)_1 (NH_3 )_{y^{ + + } } \hfill \\ (5) Co (Dien)_1 (NH_3 )_{z^{ + + } } (6) Co (Trien)_1 (NH_3 )_{a^{ + + } .} \hfill \\ \end{gathered} $$ -
5.
It has been indicated that the reduction waves of cobalt in mono- and diethanolamines can be used for the analytical estimation of the metal.
Similar content being viewed by others
References
Subrahmanya, R. S.Proc. Ind. Acad. Sci., 1956,43 A, 133.
—Ibid.,, 1956,43 A, 133.
—Ibid.,, 1957,45 A, 184.
Willis, J. B., Friend, J. A. and Mellor, D. P.J. Amer. Chem. Soc., 1945,67, 1680.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Subrahmanya, R.S. Polarographic behaviour of metals in ethanolamines. Proc. Indian Acad. Sci. 45, 195–203 (1957). https://doi.org/10.1007/BF03052722
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03052722