Summary
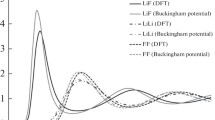
The lattice spectrum, the specific heats, and the coefficients of thermal expansion of the lithium and sodium fluorides have been calculated on the basis of the Raman dynamics of crystal vibrations, by the use of the methods developed by, the author in the earlier papers. In the case of lithium fluoride, it has been found necessary to consider the mutual repulsions of the nearest fluorine ions in addition to those existing between the neighbouring ions of lithium and fluorine. An exponential form of the repulsive potential has been assumed, the constants of which have been evaluated from the equilibrium conditions of the lattice, the-infra red absorption frequency and the compressibility. the coupling coefficients used by Kellermann have been re-evaluated to include the repulsions of the next nearest neighbours. In the case of lithium fluoride, for which adequate experimental data exist, a satisfactory agreement between the theoretical and the experimental values of the specific heats and the thermal expansion coefficients has been obtained.
Similar content being viewed by others
References
AdenstedtAnn. der Physik., 1936,26, 69.
BarnesZs. f. Physik., 1932,75, 723.
BlackmanProc. Roy. Soc. (A), 1935,149, 117.
Born and MayerZs. f. Phusik., 1932,75, 1.
BridgmanProc. Am. Acad. Arts. Sc., 1932,67 370.
DayalProc. Ind. Acad. Sci. (A), 1944), (a, b)19, 182; 224; (c)20, 24; (d, e), 77, 87;f (elsewhere in this issue).
Eucken and DannöhlZs. f. electrochem., 1934,40, 814.
GoldschmidtTrans. Farad. Soc., 1929,25, 253.
HengleinZs. f. Phys. Chem., 1925,117, 285.
HugginsJ. Chem. Phys., 1937,5, 143.
KellermannPhil. Trans. Roy. Soc. (A), 1940,283, 513.
Lyddane and HerzfeldPhys. Rev., 1938,54, 846.
ShonkaIbid., 1933,43, 947.
SlaterIbid., 1924,23, 488.
ThirringPhys. Zeits., 1914,15, 180.
Author information
Authors and Affiliations
Additional information
(Communicated by Sir C. V. Raman,kt.,f.r.s., n.l.)
Rights and permissions
About this article
Cite this article
Dayal, B. Lattice spectrum, specific heat, and thermal expansion of lithium and sodium fluorides. Proc. Indian Acad. Sci. 20, 138–144 (1944). https://doi.org/10.1007/BF03046862
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03046862