Summary
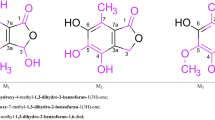
The action of mercuric acetate in methyl alcoholic solution on (1) umbelliferone, (2) 4-methylumbelliferone and (3) 4: 7-dimethylcoumarin has been investigated. With umbelliferone addition at the double bond and substitution in positions 6 and 8 take place whereas in the case of its 4-methyl derivative position 8 alone is mercurated to yield a monoacetoxymercuri-compound. In regard to the last compound the reaction stops with addition at the double bond and mercuration of the 6th position, the position 8 escaping the attack of the reagent. It is concluded that owing to the influence of the substitutent groups the reactivity of the three active centres of the coumarin molecule are differently affected.
Similar content being viewed by others
References
Seshadri and Suryaprakasa RaoProc. Ind. Acad. Sci., (A), 1936,4, 162.
PosenBer., 1881,14, 2747.
Seshadri and Suryaprakasa RaoProc. Ind. Acad. Sci., (A), 1936,3, 293.
Sen and ChakravartiJ.I.C.S., 1929, 847.
Naik and PatelJ.C.S., 1934, 1043.
Pechmann and ObermillerBer., 1901,34, 660.
Rangaswami and SeshadriProc. Ind. Acad. Sci., (A), 1937,6, 112.
ClaytonJ.C.S., 1908, 528.
Simmonis, H.Die Cumarine, p. 155.
ClaytonJ.C.S., 1910,97, 1391.
Murty, Rao and SeshadriProc. Ind. Acad. Sci., (A), 1937,6, 316.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rao, P.S., Sastri, V.D.N. & Seshadri, T.R. Reactivity of the double bond in coumarins and related unsaturated carbonyl compounds. Proc. Indian Acad. Sci. (Math. Sci.) 9, 22–28 (1939). https://doi.org/10.1007/BF03045446
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045446