Abstract
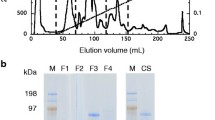
Wall-localized cellulase was partially purified from freeze-dried maize coleoptiles by a combination of DEAE-Sepharose, Superdex-200 gel filtration and Hydroxyapatite column chromatography. Activity was measured by both reducing sugar assay and dot assay on agarose gel containing carboxymethylcellulose(CMC). In situ activity staining on a nondenaturing gel overlaid on agarose gel containing CMC turned out to be a quite reliable method to detect cellulase activity. The molecular mass of partially-purified cellulase was determined to be about 53 kD based on SDS-PAGE, and the N-terminal amino acid sequence of this cellulase was NH2-AGAKGANXLGGLXRA. The enzyme hydrolyzed CMC with an optimal pH of 4.5 and optimal temperature of 40°C. It also catalyzed carboxymethylcellulose with aK m of 2.02 mg/mL and aV max of 160 ng/h/mL The β-1,4-glucosyl linkages of CMC, fibrous cellulose and lichenan were cleaved specifically by this enzyme. Reducing reagents such as cysteine-HCI, dithiothreitol and glutathione strongly enhanced the activity, suggesting that SH-groups of the enzyme were protected from oxidation. N-ethylmaleimide which is a sulfhydryl-reacting reagent did not seem to inhibit the activity, indicating that cysteine residues were not located near the active site of the enzyme. These results will be valuable in understanding the structure of wall-localized cellulase in maize coleoptiles and in predicting its possible function in the cell wall.
Similar content being viewed by others
Literature Cited
Allen RD, Lessler CI, Galewsky S, Newmann AJ (1984) The role of cellulase in hvalocysl pore formation in the mossSyrrhopodon texanus Annals ot Botany 53: 431–438
Awad M, lewis LN (1980) Avocado cellulase: extraction and purification. Food Sci 45: 1625–1628
Beguin P (1983) Detection oi cellulase activity in polyacrylamide gels using Congo red-stained agar replicas. Anal Biochem 131: 333–536
Bennett AB, Christoffersen RE (1986) Synthesis and processing of rellulase from ripening avocado fruit. Plant Physiol 81: 830–835
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quatities ot protein utilizing the printciple of protein-dye binding. Anal Hiochem 72: 248–254
Brummell DA, Lashbrook CC, Bennett AB (1994) Plant endo-1,4-β-D-glucanases: structure, properties, and physiological function.In ME. Himmel, JO Baker, RP Overend eds, tnzymalic conversion ot biomass for fuels production, ACS Symposium Series 566, pp 100–129
Byrne H, Christou NV, Verma DPS, Maclachlan GA (1975) Puritic ation and characterization of two cellulases from auxin-treated pea epicolvts. I Biol Chem 250: 1012–1018
Durbin Ml, Lewis I.N (198(5) Gellulase in Phaseolus vulgaris. Melhods in Enzymol 160: 542–551
Fry SC (1989) Cellulases, hemic elluloses and auxin-stimulated growih: a possible relationship. Physiol Plant 75: 532–530
Fry SC (1995) Polysaccharide-modifying enzymes in the plant cell wall. Annu Rev Plant Physiol Plant Mol Biol 46: 497–520
Hatfield RD, Nevins DJ (1986) Characterization of the hydrolytic activity of avocado cellulase. Plant Cell Physiol 27: 541–552
Hatfield RD, Nevins DJ (1987) Hydrolytic activity and substrate specificity of an endoglucanase fromZea mays seedling cell walls. Plant Physiol 83: 203–207
Hoson T (1993) Regulation of polysaccharide breakdown during auxin-induced cell wall loosening. J Plant Res 106: 369–381
Jung HI, Kim SI, Ha KS, Joe CO, Kang KW (1995) Isolation and characterization of guamerin, a new human leukocyte elastase inhibitor fromHirudo nipponia. J Biol Chem 270: 13879–13884
Kemmerer EC, Tucker ML (1994) Comparative study of cellulases associated with adventitious root inhibition, apical buds, and leaf, flower, and pod abscission zones in soybean. Plant Physiol 104: 557–562
Koehler DE, Lewis LN, Shannon LM, Durbin ML (1981) Purification of a cellulase from kidney bean abscission zones. Phytochemistry 20: 409–412
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 277: 680–685
Lew FT, Lewis LN (1974) Purification and properties of cellulase fromPhaseolus vulgaris. Phytochemistry 13: 1359–1366
Maclachlan G (1988) β-Clucanases fromPisum sativum. Methods in Enzymol 160: 382–391
McDougall GJ, Fry SC (1990) Xyloglucan oligosaccharides promote growth and activate cellulase: evidence for a role of cellulase in cell expansion. Plant Physiol 93: 1042–1048
Nakamura S, Mori H, Sakai F, Hayashi T (1995) Cloning and sequencing of a cDNA for poplar endo-1,4-β-glucanase. Plant Cell Physiol 36: 1229–1235
Nakamura S, Hayashi T (1993) Purification and properties of an extracellular endo-1,4-β-glucanase from suspension-cultured poplar cells. Plant Cell Physiol 34: 1009–1013
Nematollahi WPS (1996) Studies on a β-glucosidase from the cell walls of maize (Zea mays L.). Ph. D. dissertation. Univ of Texas at Austin, TX, USA
Ohmiya Y, Takeda T, Nakamura S, Sakai F, Hayashi T (1995) Purification and properties of a wall-bound endo-1,4-β-glucanase from suspension-cultured poplar cell. Plant Cell Physiol 36: 607–614
Somogyi N (1952) Notes on sugar determination. J Biol Chem 195: 19–23
Standal R, Iversen TG, Coucheron DH, Fjaervik E, Blantny JM, Valla S (1994) A new gene required for cellulose production and a gene encoding cellulolytic activity in Acetobacterxylinum are colocalized with thebcs operon. J Bacteriol 176: 665–672
Taiz L, Zeiger E (1991) Plant Physiology, Benjamin/Cummings Publishing Co, Redwood City, CA, pp 22–25
Truelsen TA, Wyndaele R (1991) Cellulase in tobacco callus: regulation and purification. J Plant Physiol 139: 129–134
Wong YS, Fincher GB, Maclachlan G (1977) Kinetic properties and substrate specificities of two cellulases from auxin-treated pea epicotyls. J Biol Chem 252: 1402–1407
Yu SC, Kim ES, Kim WK (1994) Immunocytochemical localization of cellulase on endosperm of ginseng seed. J Plant Biol 37: 435–439
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, K.S., Kim, S.H. Purification and characterization of wall-localized cellulase from maize coleoptiles. J. Plant Biol. 42, 124–132 (1999). https://doi.org/10.1007/BF03031020
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03031020