Abstract
Purpose
To determine whether prilocaine, a local anesthetic, induces apoptosis in osteoblastic cells.
Methods
After reaching subconfluence, human osteoblastic Saos-2 and MG63 cells and mouse osteoblastic MC3T3-E1 cells were exposed for 48 hr to varying concentrations of prilocaine up to 10 mM and the cytotoxicity of the cells was analyzed by phase-contrast microscopy and WST-1 assay. Saos-2 cells treated for 48 hr with 5 mM prilocaine were stained with Hoechst 33342 and nuclear fragmentation was examined under a fluorescence microscope. DNA was extracted from the cells treated with 5 mM prilocaine and DNA ladder formation (a hallmark of apoptosis) was analyzed by agarose gel electrophoresis.
Result
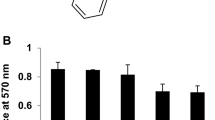
Prilocaine induced cell death in Saos-2 cells in a dose- and time-dependent manner up to the concentration of 10 mM. Marked nuclear condensation and fragmentation of chromatin were observed in the prilocainetreated cells. DNA ladder formation also was induced by prilocaine treatment. Prilocaine-induced DNA ladder formation was dose-dependent with maximal effect at a concentration of 5 mM and was time-dependent from 12 to 48 hr. DNA ladder formation was also induced by prilocaine treatment in human osteoblastic MG63 cells and mouse osteoblastic MC3T3-E1 cells. Cycloheximide prevented prilocaine-induced apoptosis in Saos-2 cells in a dose-dependent fashion up to 20 μM as determined by WST-1 assay and DNA ladder formation in agarose gel electrophoresis.
Conclusion
Osteoblastic cells treated with prilocaine exhibit both morphological and biochemical features indicative of apoptosis. The apoptotic mechanisms involve transcriptional regulation of specific proteins or protein synthesis.
Résumé
Objectif
Déterminer si la prilocaïne, un anesthésique local, a induit l’apoptose de cellules ostéoblastiques.
Méthode
Après avoir atteint le stade de sous-confluence, des cellules ostéoblastiques humaines Saos-2 et MG63 et ostéoblastiques de souris MC3T3-E1 ont été exposées pendant 48 h à des concentrations variables de prilocaïne allant jusqu’à 10 mM et la cytotoxicité des cellules a été analysée par microscopie en contraste de phase et par dosage WST-1. Les cellules Saos-2 traitées pendant 48 h avec 5 mM de prilocaïne ont été colorées avec le Hoechst 33342 et la fragmentation nucléaire a été examinée sous microscopie en fluorescence. L’ADN a été extrait et la formation d’échelle d’ADN (signe cardinal de l’apoptose) a été analysée par électrophorèse sur gel d’agarose.
Résultats
La prilocaïne a induit la mort des cellules Saos-2 d’une manière dépendante de la dose et du temps jusqu’à une concentration de 10 mM. En effet, la prilocaïne a induit dans les cellules traitées une condensation nucléaire marquée et la fragmentation de chromatine de même que la formation d’échelle d’ADN. La formation d’ADN, dépendante de la dose, a connu son effet maximal avec une concentration de 5 mM et était dépendante du temps entre 12 et 48 h. La formation d’ADN a été aussi induite par la prilocaïne dans les cellules humaines MG63 et dans les cellules de souris MC3T3-E1. La cycloheximide a empêché l’apoptose induite par la prilocaïne de se produire dans les cellules Saos-2 d’une manière dépendante de la dose pour une concentration jusqu’à 20 μM comme l’ont déterminé le dosage WST-1 et la formation d’échelle d’ADN dans électrophorèse sur gel d’agarose.
Conclusion
Les cellules ostéoblastiques traitées avec de la prilocaïne ont présenté des caractéristiques morphologiques et biochimiques indicatrices d’apoptose. Les mécanismes de l’apoptose impliquent la régulation de la transcription de protéines spécifiques ou de synthèse protéique.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Tucker GT. Pharmacokinetics of local anaesthetics. Br J Anaesth 1986; 58: 717–31.
Poste G, Allison AC. Membrane fusion. Biochim Biophys Acta 1973; 300: 421–65.
Feinstein MB, Fiekers J, Fraser C. An analysis of the mechanism of local anesthetic inhibition of platelet aggregation and secretion. J Pharmacol Exp Ther 1976; 197: 215–28.
Nash-Adler P, Louis CF, Fudyma G, Katz AM. The modification of unidirectional calcium fluxes by dibucaine in sarcoplasmic reticulum vesicles from rabbit fast skeletal muscle. Mol Pharmacol 1980; 17: 61–5.
Nicolson GL, Smith JR, Poste G. Effects of local anesthetics on cell morphology and membrane-associated cytoskeletal organization in BALB/3T3 cells. J Cell Biol 1976; 68: 395–402.
Gail MH, Boone CW. Procaine inhibition of fibroblast motility and proliferation. Exp Cell Res 1972; 73: 252–5.
Cullen BF, Haschke RH. Local anesthetic inhibition of phagocytosis and metabolism of human leukocytes. Anesthesiology 1974; 40: 142–6.
Johnson RB, Dowse CM. Comparative effects of local anesthetic preparations on gingival fibroblasts of the rat. J Dent Res 1986; 65: 1125–32.
Kingston CA, Ladha S, Manning R, Bowler K. The effect of local anaesthetics on the thermal sensitivity of HTC cells. Anticancer Res 1993; 13: 2335–40.
Wiese KG, Korabiowska M, Tymk J, Bartkowski S, Stypukowska J. The effect of lidocaine on growth of cells of head and neck squamous cell carcinoma. J Cranio-Maxillo-Fac Surg 1993; 21: 157–62.
Kim M, Lee YS, Mathews HL, Wurster RD. Induction of apoptotic cell death in a neuroblastoma cell line by dibucaine. Exp Cell Res 1997; 231: 235–41.
Kerr JFR, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972; 26: 239–57.
Wyllie AH, Kerr JFR, Macaskill LAM, Currie AR. Adrenocortical cell deletion: the role of ACTH. J Pathol 1973; 111: 85–94.
Wyllie AH. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 1980; 284: 555–6.
Rodan SB, Imai Y, Thiede MA, et al. Characterization of a human osteosarcoma cell line (Saos-2) with osteoblastic properties. Cancer Res 1987; 47: 4961–6.
Franceschi RT, James WM, Zerlauth G. 1α, 25-dihydroxyvitamin D3 specific regulation of growth, morphology, and fibronectin in a human osteosarcoma cell line. J Cell Physiol 1985; 123: 401–9.
Sudo H, Kodama H, Amagai Y, Yamamoto S, Kasai S. In vitro differentiation and calcification in a new clonal osteogenic cell line derived from newborn mouse calvaria. J Cell Biol 1983; 96: 191–8.
Takenouchi T, Munekata E. Trophic effects of substance P and ß-amyloid peptide on dibutyryl cyclic AMP-differentiated human leukemic (HL-60) cells. Life Sci 1995; 56: 479–84.
Haneji T, Kurihara N, Ikeda K, Kumegawa M. 1α, 25-dihydroxyvitamin D3 and analogues of vitamin D3 induce alkaline phosphatase activity in osteoblastic cells derived from newborn mouse calvaria. J Biochem 1983; 94: 1127–32.
Murata T, Shirakawa S, Takehara T, Kobayashi S, Haneji T. Protein phosphatase inhibitors, okadaic acid and calyculin A, induce alkaline phosphatase activity in osteoblastic cells derived from newborn mouse calvaria. Biochem Mol Biol Int 1995; 36: 365–72.
Morimoto Y, Ohba T, Kobayashi S, Haneji T. The protein phosphatase inhibitors okadaic acid and calyculin A induce apoptosis in human osteoblastic cells. Exp Cell Res 1997; 230: 181–6.
Murata T, Ansai T, Takebara T, Kobayashi S, Haneji T. Extracts ofprevotella Intermedia andActinobacillus actinomycetemcomitans inhibit alkaline phosphatase activity in osteoblastic cellsin vitro. Oral Diseases 1997; 3: 106–12.
Kerr JFR, Winterford CM, Harmon BV. Apoptosis. Its significance in cancer and cancer therapy. Cancer 1994; 73: 2013–26.
Polyak K, Xia Y, Zweier JL, Kinzler KW, Vogelstein B. A model for p53-induced apoptosis. Nature 1997; 389: 300–5.
Wiedling S. Studies on-n -propylamino-2-methylpropionanilide —a new local anaesthetic. Acta Pharmacology & Toxicology 1960; 17: 233–44.
Åström A, Persson NH. Some pharmacological properties of o-methyl—propylaminopropionanilide, a new local anaesthetic. Br J Pharmacol 1961; 16: 32–44.
Åkerman B. On felypressin (octapressin®) as an adjunct to lidocaine and prilocaine —an experimental study in animals. Acta Pharmacology & Toxicology 1966; 24: 377–88.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported in part by grants from the Grant in Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture of Japan and the grant-in-aid for cancer research from the Fukuoka Cancer Society, Fukuoka, Japan.
Rights and permissions
About this article
Cite this article
Nakamura, K., Kido, H., Morimoto, Y. et al. Prilocaine induces apoptosis in osteoblastic cells. Can J Anesth 46, 476–482 (1999). https://doi.org/10.1007/BF03012949
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03012949