Abstract
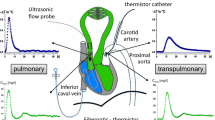
The use ofPetCO2 in detecting accidental bronchial intubation was investigated. ThePetCO2 was measured in six mongrel dogs after occluding the left mainstem bronchus in three conditions; pentobarbital anaesthesia, 0.8% halothane insufflation together withpentobarbital anaesthesia, and simultaneous left pulmonary artery and bronchial airway occlusion with intravenous pentobarbital anaesthesia. An external flow probe measured left pulmonary artery blood flow. ThePetCO2 decreased after bronchial occlusion during pentobarbital (35 ± 3 vs 30 ± 5 mmHg) and halothane-pentobarbital (30 ± 6 vs 25 ± 6 mmHg) conditions (P < 0.05). However, within three minutes of bronchial occlusion, the values ofPetCO2 had returned to their pre-occlusion values. After five minutes of bronchial occlusion pulmonary artery blood flow in the non-ventilated lung decreased (P < 0.05) during pentobarbital (770 ± 533 ml · min−1 vs 575 ± 306 ml · min−1) and halothane-pentobarbital (495 ± 127 ml · min−1 vs 387 ± 178ml · min−1) conditions. Simultaneous bronchial and pulmonary artery occlusion prevented any changes inPetCO2. It was concluded that accidental one- lung ventilation results in small and transient decreases inPetCO2. A redistribution of blood flow from the nonventilated to ventilated lung occurs which restoresPetCO2 to the original values observed with twolung ventilation.
Résumé
L’utilisation de laPetCO2 pour détecter l’intubation bronchique accidentelle a été évaluée. LaPetCO2 a été mesurée chez six chiens bâtards après avoir obstrué la bronche souche gauche sous trois conditions: une anesthésie avec pentobarbital, une anesthésie avec pentobarbital et insufflation d’halothane à 0,8%, et une anesthésie avec pentobarbital et obstruction simultanée de l’artère pulmonaire gauche et de la voie aérienne bronchique gauche. Une sonde à débit externe a été utilisée pour mesurer le débit sanguin de l’artère pulmonaire gauche. LaPetCO2 diminuait après l’obstruction bronchique lors de l’anesthésie avec pentobarbital (35 ± 3 vs 35 ± 5 mmHg) et l’anesthésie avec halothanepentobarbital (30 ± 6 vs 25 ± 6 mmHg) (P < 0,05). Cependant, moins de trois minutes après l’obstruction bronchique, les valeurs de laPetCO2 étaient revenues à leurs valeurs préocclusion. Cinq minutes après l’occlusion bronchique, les débits sanguins de l’artère pulmonaire du poumon non ventilé diminuaient (P < 0,05) durant l’anesthésie au pentobarbital (770 ± 533 ml · min−1 vs 575 ± 306 ml · min−1) et à l’halothane-pentobarbital (495 ± 127 ml · min−1 vs 387 ± 178 ml · min−1). L’occlusion simultanée de l’artère pulmonaire et de la bronche prévenait tous les changements de laPetCO2. En conclusion, la ventilation accidentelle d’un seul poumon conduit à une légère diminution transitoire de laPetCO2. Une redistribution du débit sanguin du poumon non ventilé vers le poumon ventilé se produit, ce qui rétablit laPetCO2 à sa valeur originate observée lors de la ventilation à deux poumons.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- PaCO2 (mmHg):
-
Arterial carbon dioxide pressure
- PetCO2 (mmHg):
-
End-tidal carbon dioxide pressure
- PetCO2-PetCO2 (mmHg):
-
Arterial to alveolar carbon dioxide gradient
- Qs/Qt (%):
-
Shunt fraction
- Vd/Vt:
-
Dead space ratio
References
Murray IP, Modell JH. Early detection of endotracheal tube accidents by monitoring carbon dioxide concentrations in respiratory gas. Anesthesiology 1983; 59: 344–6.
Lillie PE, Roberts JG. Carbon dioxide monitoring. Anaesth Intensive Care 1988; 16: 41–4.
Nunn JF, Hill DW. Respiratory dead space and arterial to end-tidal CO2 tension difference in anesthetized man. J Appl Physiol 1960; 5: 383–9.
Riley RH, Marcy JH. Unsuspected endobronchial intubation detection by continuous mass spectrometry. Anesthesiology 1985; 63: 203–4.
Heneghan LPH, Scallan MJH, Branthwaite MA. End-tidal carbon dioxide during thoracotomy. Anaesthesia 1981; 36: 1017–21.
Gravenstein JS, Paulus DA. Clinical Monitoring Practice. Philadephia; Lippincott Co. 1987: 193–5.
Benumof JL. Mechanism of decreased blood flow to atelectatic lung. J Appl Physiol 1979; 46: 1047–8.
Rossing RG, Cain SM. A nomogram relating PO2, pH, temperature, and hemoglobin saturation in the dog. J Appl Physiol 1966; 21: 195–201.
Severinghaus JW. Water vapor calibration errors in some capnometers: respiratory conventions misunderstood by manufacturers? Anesthesiology 1989; 70: 996–8.
Fletcher R, Johnson B. Dead space and single breath test for carbon dioxide during anaesthesia and artificial ventilation. Br J Anaesth 1984; 56: 109–19.
Nunn JF. Applied Respiratory Physiology, 2nd ed. London, England: Butterworths. 1981: 213–8.
Sokol RR, Rohlf FJ. Biometry Statistical Tables. San Francisco: Will Freenan and Co. 1981; 81.
Chang PC, Reynolds FB, Lang SA, Ha HC. Endobronchial intubation in dogs. Can J Anaesth 1990; 37: 287–95.
Barer GR, Howard P, McCurrie JR, Shaw JW. Changes in the pulmonary circulation after bronchial occlusion in anesthetized dogs and cats. Circ Res 1969; 25: 747–64.
Wilson LB, Levitz KY. Chemoreflex blunting of hypoxic pulmonary vasoconstriction is locally mediated. J Appl Physiol 1989; 66: 722–91.
Bjertnaes LJ. Hypoxia induced vasoconstriction in isolated perfused lungs exposed to injectable or inhalation anesthesia. Acta Anaesthesiol Scand 1977; 21: 133–47.
Russell GB, Graybeal JM, Strout JC. Stability of arterial to end-tidal carbon dioxide gradients during postoperative cardiorespiratory support. Can J Anaesth 1990; 5: 560–6.
Author information
Authors and Affiliations
Additional information
This work was supported by grants from the Saskatchewan Health Research Board and the Canadian Heart and Stroke Foundation.
Rights and permissions
About this article
Cite this article
Johnson, D.H., Chang, P.C., Hurst, T.S. et al. Changes inPetCO2 and pulmonary blood flow after bronchial occlusion in dogs. Can J Anaesth 39, 184–191 (1992). https://doi.org/10.1007/BF03008654
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03008654