Abstract
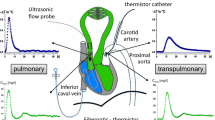
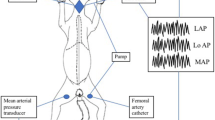
Using an isolated lung the effects of halothane on hypoxic pulmonary vasoconstriction (HPV) were studied in the presence of cyclooxygenase blockade. The pulmonary vasculature can be divided into arterial, middle and venous segment resistances. Analysis of the vascular pressure-flow relationship further separates resistance into a flow dependant resistance (1/slope) and a zero-flow pressure intercept (Pcrit). We ventilated six lobes with control (35 per cent O2) and hypoxic (three per cent O2) gas mixtures with the addition of either 0, 0.5, 1.0, or 2.0 per cent halothane. We found that after addition of indomethacin (5 mg · kg− 1), ventilation with three per cent O2 increased total resistance by 87 per cent over baseline with the increase primarily in the middle vascular segment. During normoxic ventilationPcrit was 7.9 cm H2O and this increased significantly with hypoxia to 11.5 cm H2O). Only 2.0 per cent halothane blocked the increases in middle segment resistance and inPcrit. We conclude that following cyclooxygenase blockade, halothane inhibits HPV by acting on middle segment vessels.
Résumé
Nous avons étudié, sur poumon isolé, les effets de l’halothane sur la vasoconstriction pulmonaire hypoxique (HPV) en présence d’inhibiteur de la cyclooxygénase. Les secteurs artériel, médian et veineux contribuent successivement à la résistance vasculaire pulmonaire. En analysant les courbes pressiondébit, on peut calculer la résistance (1/pente) et la pression d’ouverture (PCRIT). NOUS ventilions six lobes pulmonaires avec de I’oxygène à 35 pour cent (contrôle) ou à trois pour cent (hypoxie) en y ajoutant 0, 0,5, 1,0 ou 2,0 pour cent d’halothane. Après l’injection de 5 mg · kg− 1 d’indométhacine et avec trois pour cent d’O2, la résistance totale augmentait de 87 pour cent par rapport au contrôle, surtout en secteur médian. LaPcrit était significativement moins élévee en condition normoxique, passant de 7,9 à 11,5 cmH2O en condition hypoxique. Il fallait deux pour cent d’halothane pour bloquer cette augmentation de resistance et de pression d’ouverture. Done, malgré une inhibition de la cyclooxygénase, l’halothane renverse l’HPV du secteur vasculaire médian.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Loh L, Sykes MK, Chakrabarti MK. The effects of halothane and ether on the pulmonary circulation in the innervated perfused cat lung. Br J Anaesth 1977; 49: 309–13.
Heitz DC, Jelbson PJ, Boutros AR, Brody MJ. The effects of halothane on the pulmonary vascular bed of the dog. Anesthesiology 1977; 35: 61–7.
Marshall C, Kim SD, Marshall BE. The actions of halothane, Ibuprofen, and BW 755C on hypoxic pulmonary vasoconstriction. Anesthesiology 1987; 66: 537–42.
Voelkel NF, Gerber JG, McMurty IF, Nies AS, Reeves JT. Release of vasodilator prostaglandins PGI2 from isolated rat lung during hypoxia. Circ Res 1981; 48: 27–213.
Hakim TS, Michel RP, Minami H, Chang HK. Site of hypoxic vasoconstriction studied with arterial and venous occlusion. J Appl Physiol 1983; 54: 1298–302.
Rock P, Patterson GA, Permut S, Sylvester JT. Nature and distribution of vascular resistance in hypoxic pig lungs. J Appl Physiol 1985; 59: 1892–901.
Hakim TS, Michel RP, Chang HK. Partitioning of pulmonary vascular resistance in dogs by arterial and venous occlusion. J Appl Physiol 1982; 52: 710–5.
Johnson D, Mayers I, To T. The effects of halothane in hypoxic pulmonary vasoconstriction. Anesthesiology 1990; 72: 125–33.
Burnett RW, Noonan DC. Calculations and correction factors used in determination of blood PH and blood gases. Clin Chem 1974; 20: 1499–506.
Rossing RG, Cain SM. A nomogram relating PO2, PH, temperature and hemoglobin saturation in the dog. J Appl Physiol 1966; 21: 195–201.
Snedecor GW, Cochran WG. Statistical methods. 7th ed. Iowa: The Iowa State University Press, 1980.
Sokal RR, Rohlf FJ. Biometry. San Francisco: W.H. Freeman and Co., 1981.
Townsley MI, Korthus RJ, Taylor AE. Effects of arachidonatc on permeability and resistance distribution in canine lungs. J Appl Physiol 1985; 58: 206–10.
Rudolph AM, Yuan S. Response of the pulmonary vasculature to hypoxia and H+ ion concentration. J Clin Invest 1966; 45: 399–411.
Julien M, Hakim TS, Vahi R, Chang HK. Effect of hematocrit on vascular pressure profile in dogs’lungs. J Appl Physiol 1985; 58: 743–8.
Mayers I, Johnson DH, Wobeser W, To T. The effects of indomethacin in hypoxic pulmonary vasoconstriction in canine lobes. Am Rev Resp Dis 1989; 139: A617.
Hales CA, Rouze E, Buchwals IA, Kazemc H. Role of prostaglandins in alveolar hypoxic vasoconstriction. Resp Physiol 1977; 29: 151–62.
Rubin LJ, Lazar JD. Influence of prostaglandin synthesis inhibitors on pulmonary vasodilator effects of hydralazine in dogs with hypoxic pulmonary vasoconstriction. J Clin Invest 1981; 67: 193–200.
Vaage J, Bjertnaes LL, Hauge A. The pulmonary vasoconstrictor response to hypoxia: effects of inhibitors of prostaglandin biosynthesis. Acta Physiol Scand 1975; 95: 95–101.
Hales CA, Rouse ET, Slate JL. Influence of aspirin and indomethacin on variability of alveolar vasoconstriction. J Appl Physiol 1978; 45: 33–9.
Versprile A. Pulmonary vascular resistance. Intensive Care Med 1984; 10:51–3.
Graham R, Skoog C, Oppenheimer L, Rabson J, Goldberg H. Critical closure in the canine pulmonary vasculature. Circ Res 1982; 50: 566–72.
Murray PA, Lodato RF, Michael JR. Neural antagonists modulate pulmonary vascular pressure-flow plots in conscious dogs. J Appl Physiol 1986; 60: 1900–7.
Author information
Authors and Affiliations
Additional information
Supported by the ICI Pharma Canadian Research Award in Anaesthesia. Also supported by a grant from the Saskatchewan Heart Foundation.
Rights and permissions
About this article
Cite this article
Johnson, D., Mayers, I. & Hurst, T. Halothane inhibits hypoxic pulmonary vasoconstriction in the presence of cyclooxygenase blockade. Can J Anaesth 37, 287–295 (1990). https://doi.org/10.1007/BF03005577
Issue Date:
DOI: https://doi.org/10.1007/BF03005577