Abstract
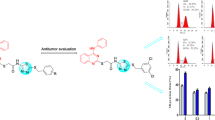
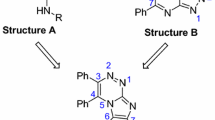
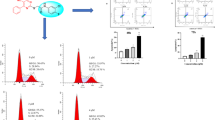
Seven isoazamitosene derivatives, mitomycin analogues, were synthesized and tested for cytotoxicities against leukemia and gastric cancer cell lines. Preparation of a pyrrolo[1,2-a]benzimidazole (3) (azamitosene ring system) was completed by utilizing the Lewis acid-catalized cyclization, witho-chloronitrotoluene as the starting material. Nitration of3 produced a mixture of two isomers (5-nitro isomer (4) and 7-nitro isomer (5)) in product ratio of 36∶52.4 was directly converted into quinone (7) by reduction and Fremy oxidaton. Finally, quinone derivatives (8, 9, 10, and11) were synthesized by 1,4-addition of7 with cyclic secondary amines. From above-mentioned5, 8-nitro compound (15) was prepared in 4 steps. At pH 3, Fremy oxidation of15 produced quinone (16), whereas iminoquinone derivatives (17a and17b) at pH 7. Isoazamitosene derivatives (8, 9, 10, and11), containing cyclic amino groups at the 7-position, showed potent cytotoxicity on P388, SNU-1, and KHH tumor cell lines. Among them,8 had stronger cytotoxicity against SNU-1 cell line than mitomycin and adriamycin. Considering these results, isoazamitosene derivatives may had unique cytotoxicity profiles. However, isoiminoazamitosene derivatives (17a and17b) revealed very weak cytotoxicity.
Similar content being viewed by others
References Cited
Andrews, P.A., Pan, S. and Bachur, N.R., Electrochemical reductive activation of mitomycin C.J. Am. Chem. Soc., 108, 4158–4166 (1986).
Begleiter, J.H., Cytocidal action of the quinone group and its relationship to antitumor activity.Cancer Res., 43(2), 481–484 (1983).
Carmichel, J., DeGraff, W.G. and Gazdar, A.F., Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing.Cancer Res., 47, 939–944 (1986).
Doroshow, J.H., Effect of anthracycline antibiotics on oxygen radical formation in rat heart.Cancer Res., 43(2), 460–472 (1983).
Fishbein, P.L., Synthesis and antineoplastic activity of 1a-formyl and 1a-thioformyl derivatives of mitomycin C and 2-methylaziridine.J. Med. Chem., 30, 1767–1773 (1987).
Islam I. and Skibo E.B., Synthesis and physical studies of azamitosene and iminoazamitosene reductive alkylating agents. Iminoquinone hydrolytic stability, syn/anti isomerization, and electrochemistry.J. Org. Chem., 55, 3195–3205 (1990).
Islam I. and Skibo E.B., Structure-activity studies of antitumor agents based on pyrrolo[1,2-a] benzimidazoles: new reductive alkylating DNA cleaving agents.J. Med. Chem., 34, 2954–2961 (1991).
Iyengar, B.S., Sami, S.M., Remers, W.A., Bradner, W.I. and Schurig, J.E., Mitomycin C and porfiromycin analogues with substituted ethylamines at position 7.J. Med. Chem., 26, 16–20 (1983).
Kennedy, K.A., Teicher, B.A., Rockwell, S. and Sartorelli, A.C., The hypoxic tumor cell: a target for selective cancer chemotherapy.Biochem. Pharm., 29, 1–8 (1980).
Keyes, S.R., Heimbrook, D.C., Fracasso, P.M., Rockwell, S., Sligar, S.G. and Sartorelli, A.C., Chemotherapeutic attack of hypoxic tumor cells by the bioreductive alkylating agent mitomycin C.Adv. Enz. Reg., 23, 291–307 (1985).
Math-Cohn, O. and Suschitzky, H., Heterocycles by ring closure of ortho-substituted t-anilines (the t-amino effect),Adv. Heterocycl. Chem., 14, 178–211 (1972).
Moore, H.W., Bioactivation as a model for drug design bioreductive alkylation.Science, 197, 527–532 (1977).
Moore, H.W. and Czerniak, P., Naturally occurring quinones as potential bioreductive alkylating agents.Med. Res. Rev., 1, 249–280 (1981).
Perrin, D.D., Armarego, L.F. and Perrin, O.R.,Purification of Laboratory Chemicals.2nd ed. Pergamon Press, New York, 1980.
Sami, S.M., Iyengar, B.S., Tarnow, S.E., Remer, W.A., Bradner, W.T. and Schurig, J.E., Mitomycin C analogues with aryl substitutions on the 7-amino group.J. Med. Chem., 27, 701–708 (1984).
Skibo, E.B. and Schulz W.G., Pyrrolo[1,2-a]benzimidazole-based aziridinyl quinones. a new class of DNA cleaving agent exhibiting G and A Base specificity.J. Med. Chem., 36, 3050–3055 (1993).
Tomasz, M., Lipman, R., Verdine, G.L. and Nakanishi, K., Reassignment of the guanine-binding mode of reduced mitomycin C.Biochemistry, 25, 4337–4344 (1986).
Vyas, D.M., Chiang, Y., Benigni, D. and Doyle, T.W., Novel mitomycin C amidines: Synthesis and their reactions with amines.J. Org. Chem., 52, 5601–5605 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ahn, C.M., Kim, S.K. Synthesis andin vitro antitumor activity of isoazamitosene and isoiminoazamitosene derivatives. Arch. Pharm. Res. 19, 535–542 (1996). https://doi.org/10.1007/BF02986024
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02986024