Abstract
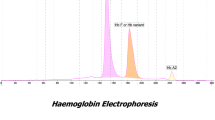
We describe a 6-year-old girl and her mother with dominant β-thalassemia due to hemoglobin Hradec Kralove (Hb HK). Both patients presented microcytic anemia, jaundice, splenomegaly, cholelithiasis, and recurrent hemolytic bouts. Osmotic resistance tests using saline and coiled planet centrifugation revealed the increased fragility of the red cell membrane. On the other hand, the glycerol lysing time was prolonged, and results of the isopropanol test were weakly positive. Despite mimicking the features of hereditary spherocytosis, the results of the genetic analyses verified the second reported family with Hb HK (codon 115,\(G\underset{\raise0.3em\hbox{$\smash{\scriptscriptstyle-}$}}{C} C[Ala] \to G\underset{\raise0.3em\hbox{$\smash{\scriptscriptstyle-}$}}{A} C[Asp]\)). Splenectomy was effective for the amelioration of hemolysis. Of 7 reported patients with Hb variants at β-globin codon 115 (Hb Madrid and Hb HK), 5 underwent splenectomy. Because of the variable augmentation of extramedullary hemolysis in dominant β-thalassemias, genotyping is necessary for determining the clinical indication of splenectomy.
Similar content being viewed by others
References
Forget BG. The β-thalassemias. In: Steinberg MH, Forget BG, Higgs DR, Nagel RL, eds.Disorders of Hemoglobin, Genetics, Pathophysiology, and Clinical Management. Cambridge, UK: Cambridge University Press; 2001;231–388.
Olivieri NF. The beta-thalassemias.N Engl J Med. 1999;341:99–109.
Adams JG 3rd, Coleman MB. Structural hemoglobin variants that produce the phenotype of thalassemia.Semin Hematol. 1990;27:229–238.
Thein SL. Dominant beta thalassaemia: molecular basis and pathophysiology.Br J Haematol. 1992;80:273–277.
Kazazian HH Jr, Orkin SH, Boehm CD, et al. Characterization of a spontaneous mutation to a beta-thalassemia allele.Am J Hum Genet. 1986;38:860–867.
Ohba Y. Unstable hemoglobins.Hemoglobin. 1990;14:353–388.
Thein SL. Is it dominantly inherited beta thalassaemia or just a beta-chain variant that is highly unstable?Br J Haematol. 1999;107:12–21.
Advani R, Sorenson S, Shinar E, Lande W, Rachmilewitz E, Schrier SL. Characterization and comparison of the red blood cell membrane damage in severe human alpha- and beta-thalassemia.Blood. 1992;79:1058–1063.
Wakamatsu C, Ichinose M, Manabe J, et al. Molecular basis of betathalassemia in Japan: heterogeneity and origins of mutations.Acta Haematol. 1994;91:136–143.
Chifu Y, Nakashima H, Hara T, Yokota E, Imamura T. Betathalassemia major resulting from a compound heterozygosity for the beta-globin gene mutation: further evidence for multiple origin and migration of the thalassemia gene.Hum Genet. 1992;89:343–346.
Kitajima K, Shibata S. Coiled planet centrifugation and its application to the observation of altered membrane properties of erythrocytes in hepatobiliary disorders.J Lab Clin Med. 1975;85:855–864.
Outeirino J, Casey R, White JM, Lehmann H. Haemoglobin Madrid beta 115 (G17) alanine?proline: an unstable variant associated with haemolytic anaemia.Acta Haematol. 1974;52:53–60.
Divoky V, Svobodova M, Indrak K, Chrobak L, Molchanova TP, Huisman TH. Hb Hradec Kralove (Hb HK) or alpha 2 beta 2 115(G17)Ala?Asp, a severely unstable hemoglobin variant resulting in a dominant beta-thalassemia trait in a Czech family.Hemoglobin. 1993;17:319–328.
Molchanova TP, Postnikov YuV, Wilson JB, et al. Hb Madrid or alpha 2 beta (2)115 (G17)Ala?Pro in a black teenager.Hemoglobin. 1993;17:251–254.
Maehara T, Tsukamoto N, Nojima Y, et al. Enhanced haemolysis with beta-thalassaemia trait due to the unstable beta chain variant, Hb Gunma, accompanied by hereditary elliptocytosis due to protein 4.1 deficiency in a Japanese family.Br J Haematol. 2002;117:193–197.
Honig GR, Shamsuddin M, Zaizov R, Steinherz M, Solar I, Kirschmann C. Hemoglobin Petah Tikva (alpha 110 ala replaced by asp): a new unstable variant with alpha-thalassemia-like expression.Blood. 1981;57:705–711.
Kazazian HH Jr, Dowling CE, Hurwitz RL, Coleman M, Stopeck A, Adams JG 3rd. Dominant thalassemia-like phenotypes associated with mutations in exon 3 of the beta-globin gene.Blood. 1992;79:3014–3018.
Beris P, Miescher PA, Diaz-Chico JC, et al. Inclusion body betathalassemia trait in a Swiss family is caused by an abnormal hemoglobin (Geneva) with an altered and extended beta chain carboxyterminus due to a modification in codon beta 114.Blood. 1998;72:801–805.
Fucharoen S, Fucharoen G, Fukumaki Y, et al. Three-base deletion in exon 3 of the beta-globin gene produced a novel variant (beta Gunma) with a thalassemia-like phenotype.Blood. 1990;76:1894–1896.
Fucharoen S, Kobayashi Y, Fucharoen G, et al. A single nucleotide deletion in codon 123 of the beta-globin gene causes an inclusion body beta-thalassaemia trait: a novel elongated globin chain beta Makabe.Br J Haematol. 1990;75:393–399.
Giordano PC, Harteveld CL, Michiels JJ, et al. Phenotype variability of the dominant beta-thalassemia induced in four Dutch families by the rare cd121 (G?T) mutation.Ann Hematol. 1998;77:249–255.
Van Weel M, Harteveld CL, Bernini LF, Brouwers TM, Giordano PC. The second case of dominant beta-thalassemia induced by the codon 127 (CAG?TAG) described as a de novo mutation in a Dutch boy.Hemoglobin. 1999;23:389–393.
Hall GW, Thein S. Nonsense codon mutations in the terminal exon of the beta-globin gene are not associated with a reduction in betamRNA accumulation: a mechanism for the phenotype of dominant beta-thalassemia.Blood. 1994;83:2031–2037.
Ho PJ, Hall GW, Watt S, et al. Unusually severe heterozygous betathalassemia: evidence for an interacting gene affecting globin translation.Blood. 1998;92:3428–3435.
Eldor A, Durst R, Hy-Am E, et al. A chronic hypercoagulable state in patients with beta-thalassaemia major is already present in childhood.Br J Haematol. 1999;107:739–746.
Eldor A, Rachmilewitz EA. The hypercoagulable state in thalassemia.Blood. 2002;99:36–43.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ohga, S., Nomura, A., Takada, H. et al. Dominant β-Thalassemia with Hemoglobin Hradec Kralove: Enhanced Hemolysis in the Spleen. Int J Hematol 78, 329–334 (2003). https://doi.org/10.1007/BF02983557
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02983557