Abstract
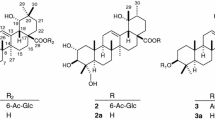
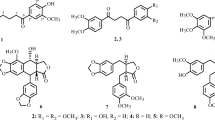
Activity-guided fractionation of the ethanol extract of the whole plant fromSolanum lyratum resulted in the isolation of a new pregnane derivative glycoside, 16-dehydropregnenolone 3-O-α-l-rhamnopyranosyl-(1→2)-β-d-glucopyranosid uronic acid (2), as well as other six known compounds: 16-dehydropregnenolone (1), allopregenolone (3), protocatechuic acid (4), vanillic acid (5), caffeic acid (6), and scopoletin (7). The structure of the isolated compounds were elucidated on the basis of their spectral data and chemical evidences. Compounds1, 3, 4 were isolated for, the first time from this plant. Cytotoxic activities of the isolated compounds were evaluated. Compound1 exhibited significant cytotoxic activity against A375-S2, HeLa, SGC-7901, and Bel-7402 with IC50 values of 13.1±0.9, 21.5±1.0, 40.2±0.7, and 49.8±1.2 μg/mL, respectively.
Similar content being viewed by others
References
Dawe, R. D. and Wright, L. C., An improved route to pregna-5,20-dien-3β-ol(muricin aglycone): Carbon and proton nuclear magnetic resonance assignments for the aglycone and a number of related pregnant derivatives.Can. J. Chem., 65, 666–669 (1987).
Kang, S. Y., Sung, S. H., Park, J. H., and Kim, Y. C., Hepatoprotective activity of scopoletin, a constituent ofSolanum Lyratum.Arch. Pharm. Res., 21, 718–722 (1998).
Mosmann, T., Rapid colorimetric assay for celluar growth and survival: Application to proliferation and cytotoxicity assays.J. Immunol. Methods, 65, 55–63 (1983).
Murakami, K., Ezima, H., and Takaishi, Y., Studies on the constituents of solanum plants. v: The constituents ofS. lyratum Thunb. II.Chem. Pharm. Bull., 33, 67–73 (1985).
Park, S. W., Yook, C. S., and Lee, H. K., Chemical components from the fruits ofCrataegus pinnatifida var.psllosa.Kor. J. Pharmacogn., 25, 328–335 (1994).
Park, W. Y., Lee, S. C., Ahn, B. T., Lee, S. H., Ro, J. S., and Lee, K. S., Phenolic compounds fromAcalypha australis L.Kor. J. Pharmcogn., 24, 20–25 (1993).
Shan, C. M., Hu, J. J., and Du, G. H., Apoptosis of hepotoma cell line BEL-7404 induced by extracts of Solanum lyratum Thunb.Chinese Journal Of Clinical Pharmacology and Therapeutics, 6, 200–203 (2001).
Shi, W. R. and Yan, L., Influence ofSolanum Lyratum Thumb on HL-60.J. of Fujian College of TCM, 12, 36–38 (2002).
Steck, W. and Mazurek, M., Identification of natural coumarins by nmr spectroscopy.Lioydia, 35, 418–439 (1972).
Yahara, S., Morooka, M., and Ikeda, M., Two new steroidal glucuronides fromSolanum lyratum II.Planta medica, 52, 496–498 (1986).
Yang, J. Z., Guo, G. M., Zou, L. X., and Ding, Y., Studies on chemical constituent ofSolanum Lyratum.ZhongGuo Zhongyao Zazhi, 27, 42–43 (2002).
Yahara, S., Murakami, N., and Yamasaki, M., A furostanol glucoronide fromSolanum lyratum.Phytochemistry, 24, 2748 (1985).
Yung, Y. L., Feng, L. H., and Toshihiro, N., Two new soladulcidine glycosides fromSolanum lyratum.Chem. Pharm. Bull., 45, 1381–1382 (1997).
Zsuzsa, S., Peter, F., and Frederick, S., Complete1H and13C NMR spectra of pregnenolone.Steroids, 60 442–446 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, LX., Fu, Ww., Ren, J. et al. Cytotoxic constituents fromSolanum lyratum . Arch Pharm Res 29, 135–139 (2006). https://doi.org/10.1007/BF02974274
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02974274