Abstract
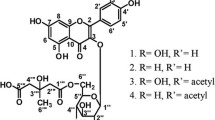
A new flavanone Persinol (1) and the new flavanone glucosides persinosides A (2) and B (3) along with known flavonoids (4 and5) have been isolated from the ethyl acetate soluble fraction of the whole plants ofAerva persica. Their structures were elucidated on the basis of extensive analysis of nuclear magnetic resonance (1D & 2D-NMR) spectral data. All of them showed profound antioxidative activities by DPPH and cytochrome-c-reduction assays using the HL-60 cell culture system.
Similar content being viewed by others
References
Arora, A., Nair, M. G., and Strasburg, G. M., Structure activity relationships for antioxidant activities of a series of flavonoids in a liposomal system.Free Radical Biological Medicines, 24, 1355–1363 (1998).
Cao, G., Sofic, E., and Prio, R. L., Antioxidant and prooxidant behaviour of flavonoids structure-activity relation.Free radical Biological Medicine, 22, 749–760 (1997).
Forkman, G., 5,7,3′,4′,5′-Pentahydroxyflavanone in the bracts ofHelichrysum bracteatum.Z. Naturforch. C, 38, 891–93 (1983).
Gaffield, W., Circular dichroism, optical rotatory dispersion and absolute configuration of flavanones, 3-hydroxyflavanonones and their glycosides: Determination of agylcone chirality in flavanone glycosides.Tetrahedron, 26, 4093–4108 (1970).
Garg, S. P., Bhushan, R., and Kapoor, R. C., Chrysin-7-O-galactoside: a new flavonoid fromAerva persica.Indian. J. Chem., 17B, 416–417 (1979).
Garg, S. P., Bhushan R., and Kapoor, R. C., Aervanone, a new flavonoid fromAerva persica.Phytochemistry, 19, 1265–1267 (1980).
Gonzalez, A. G., Bernejo, J., Estevez, F., and Velazquez, R., Phenolic derivatives fromArtemisia glutinosa.Phytochemistry, 22, 1515–1516 (1983).
Gorin, P. A. J. and Mazurek, M., Further studies on the assignment of signals in13C magnetic resonance spectra of aldoses and derived methyl glycosides.Can. J. Chem., 53, 1212–1223 (1975).
Gujer, R., Magnolato, D., and Self, R., Glucosylated flavonoids and other phenolic compounds fromSorghum.Phytochemistry, 25, 1431–1436 (1986).
Horowitz, R. M. and Gentili, B., Flavonoids of citrus. IV. Isolation of some aglycones from the lemon (Citrus limon).J. Org. Chem., 25, 2183–2187 (1960).
Mabry, T. J., Markham, K. R., and Thomas, M. B., “The systematic identification of flavonoids”, Springer-Verlag, New York, pp 140–147 (1970).
Mabry, T. J., Markham, K. R., and Thomas, M. B., “The systematic identification of flavonoids”, Springer-Verlag, New York, pp. 267–268 (1970).
Mabry, T. J. and Markham, K. R., “The Flavonoids”, Champman and Hall, London, pp. 79–80 (1975).
Oliveira, A. B., Fonseca, S. L. G., and Gottlieb, O. R., Flavonoids and coumarins fromPlatymiscium praecox.Phytochemistry, 11, 3515–3519 (1972).
Pfeffor, P. E., Valentine, K. M., and Parrish, F. W., Deuterium-induced differential isotope shift13C NMR. 1. Resonance Reassignments of mono-and disaccharides.J. Am. Chem. Soc., 101, 1265–1274 (1979).
Rani, I., Structural confirmation of eriodictyol 7-methyl ether, a new flavanone occurring inWyethia glabra Nutt.Indian J. Chem. Sect B, 25, 1251–1252 (1986).
Santzke, G. and Znatzke, F., Chiral properties of 4-substituted flavans.Tetrahedron, 29, 909–912 (1973).
Sharma, S., Stutzman, J. D., Kelloff, G. J., and Steele, V. E., Screening of potential chemopreventive agents using biochemical markers of carcinogenesis.Screening of Cancer Research, 54, 5848–5855 (1994).
Susan, Mc. C., Kathleen, H., and Bruce, B., Flavonoids fromWyethia glabra.Phytochemistry, 24, 1614–1416 (1985).
Wollenweber, E., Epicuticular leaf flavonoids fromEucalyptus species andKalmia latifolia.Z. Natureforch., C, 36, 913–915 (1981).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmed, E., Imran, M., Malik, A. et al. Antioxidant activity with flavonoidal constituents fromAerva persica . Arch Pharm Res 29, 343–347 (2006). https://doi.org/10.1007/BF02968582
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02968582