Abstract
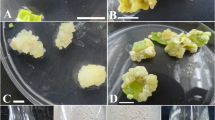
This study was conducted to establish a plant cell culture system for the production of medically important secondary metabolites fromXanthium strumarium. The effects of plant growth regulators including NAA, 2,4-D, kinetin, and ABA were examined in terms of callus induction, maintenance of callus and suspension cultures. It was shown that callus was induced upon treatment with NAA while embryo was induced after treatment with 2,4-D. Callus formation was further improved by treatment with ABA and NAA. The level of callusing increased by 17–29% for the seed case, cotyledon, leaf, and hypocotyl and by 96% in the case of the root. Suspension cell lines were established using calli produced from cotyledon, hypocotyl and root and cultured at 25°C under light conditions. The cells grew up to 15 g/L with NAA 2 ppm, BA 2 ppm, and ABA 1 ppm treatment. Supernatants of suspension cultures of cell lines derived from coyledon and hypocotyl produced some distinctive secondary metabolites, one of which was identified as 8-epi-tomentosin, which belongs to the xanthanolides. The amounts of 8-epi-tomentosin produced by the cotyledon-and hypocotylderived cell lines were 13.4 mg/L and 11.0 mg/L, respectively.
Similar content being viewed by others
References
Wink, M. (1998) Plant breeding: Importance of plant secondary metabolites for protection against pathogens and herbivores.Theor. Appl. Genet. 75: 225–233.
Agata, I., S. Goto, T. Hatano, S. Nishibe, and T. Okuda (1993) 1,3,5-tri-O-Caffeoylquinic acid fromXanthium strumarium.Phytochem. 33: 508–509.
Chopra, R. N., I. C. Chopara, K. L. Handa, and L. D. Kapur (1958)Indigeous Drugs of India. 2nd ed. U. N. Dhar and Sons, Calcutta, India.
Lee T. B. (1979)Illustrated Flora of Korea. p. 733. Academy Press, Seoul, Korea.
Talakai, T. S., S. K. Dwivedi, and S. R. Sharma (1995)In vitro andin vivo antitrypanosomal activity ofXanthium strumarium leaves.J. Ethnopharm. 49: 141–145.
Lee, D. B. (1974) Illustrated encyclopedia of fauna of Korea.Economic Plants 15: 423.
Mukherji, S., A. K. Banerjee, and B. M. Mitra (1970) Plant antitumour agents.Indian J. Pharmacy 32: 48–49.
Dhar, M. L., M. M. Dhar, B. N. Dhawan, B. N. Mehrotra, and C. Ray (1968) Screening of indian plants for biological activity: Part I.Indian J. Exp. Biol. 6: 232–247.
Kim, H. S. and J. O. Shin (1997) Isolation and antimicrobial activity ofXanthium strumarium L.Kor.J. Appl. Microbiol. Biotechnol. 25: 183–188.
Ellis, J. P. and N. D. Camper (1994)In vitro culture ofXanthium strumarium (Cocklebur).Plant Cell Tiss. Org. 36: 369–372.
Ellis, J. P. and N. D. Camper (1995)In vitro cultured cocklebur (Xanthium strumarium L.) response to dimercapto-propanesulfonic acid and monosodium methanearsonate.J. Plant Growth Regulation 14: 9–13.
Ammirato, P. V. (1983)Handbook of Plant Cell Culture, Technique for Propagation and Breeding Vol. 1. D. A. Eavns, W. R. Sharp, P. V. Ammirato and Y. Yamada (eds.). Mac-Millan Pub. Co., NY, USA.
Ahn, I. W., Z. S. No, S. Y. Ryu, O. P. Zee, and S. K. Kim (1995) Isolation of cytotoxic compounds from the leaves ofXanthium strumarium L.Natural Product Sci. 1: 1–4.
Joshi, S. P., S. R. Rojatkar, and B. A. Nagasampag (1997) Antimalarial activity ofXanthium strumarium.J. Med. Plant Sci. 19: 366–368.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, J.S., Yi, G.H., Nam, M.H. et al. Production of 8-epi-tomentosin by plant cell culture ofXanthium strumarium . Biotechnol. Bioprocess Eng. 6, 51–55 (2001). https://doi.org/10.1007/BF02942250
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02942250