Abstract
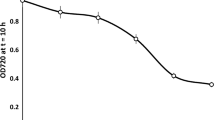
Brewery bottom yeast strain 95 from thePilsner Urquell propagation unit was used to reappraise the efficiency of the acidification power (AP) test consisting in determining the spontaneous (oxygen-induced) and glucose-induced medium acidification caused by yeast and lactic acid bacteria under standard conditions, and used widely for assessing and predicting the vitality of industrial strains. AP was evaluated in yeast stored for different periods of time (0–28 d) at 4 °C, at different temperatures before and during the test (0–55 °C), and at different concentrations of cells and glucose and different cells-to-glucose ratios. All these factors had a strong effect on acidification kinetics and the AP value. By contrast, the duration of the lag period between yeast collection and the test (0–6 h) had no perceptible effect on the AP value. The best results were achieved at saturation concentrations of cells (>10 g pressed yeast or ≈14 g yeast slurry per 100 mL) and glucose (≈3 %) and at 25 °C. Since an exact evaluation of acidification characteristics depends strongly on the kinetics of the process, the AP test should include monitoring the time course of the acidification.
Similar content being viewed by others
Abbreviations
- AP:
-
acidification power
- APmin :
-
acidification power calculated with the aid of ΔpHsp min
- ΔpH10 :
-
pH change after 10 min
- ΔpH20 :
-
pH change after 20 min
- ΔpHsp :
-
spontaneous pH change after suspending yeast in water
- ΔpHGlc :
-
Glc-induced pH change
- YATA:
-
yeast acidification and turbidity analyzer
- ΔpHmin :
-
pH change at the moment of strongest acidification (attainment of lowest pH value)
- ΔpHsp min :
-
pH change at the moment of the strongest spontaneous acidification (attainment of the lowest pH value)
References
Collar C., Bollain C., Angioloni A.: Significance of microbial transglutaminase on the sensory, mechanical and crumb grain patterns of enzyme supplemented fresh pan breads.J.Food Engn. 70, 479–488 (2005).
Fernandez S.S., Gonzales G., Sierra J.A.: The acidification power test and the behavior of yeast in brewery fermentations.Tech. Quart.Master Brew.Assoc.Am. 28, 28–95 (1991).
Fernandez S.S., Gonzalez M.G., Sierra J.A.: Evaluation of the effect of acid washing on the fermentative and respiratory behavior of yeasts by the acidification power test.Tech.Quart.Master Brew.Assoc.Am. 30, 1–8 (1993).
Gatto E., Peddie F., Andrews S.: Acidification power: performance evaluation of freeze-dried lactic acid bacteria.Food Australia 45, 124–128 (1993).
Higgins V.J., Bell P.J.L., Dawes I.A., Attfield P.V.: Generation of a novelSaccharomyces cerevisiae strain that exhibits strong maltose utilization and hyperosmotic resistance using nonrecombinant techniques.Appl.Environ.Microbiol. 67, 4346–4348 (2001).
Imai T., Nakajima I., Ohno T.: Development of a new method for evaluation of yeast vitality by measuring intracellular pH.Tech. Quart.Master Brew.Assoc.Am. 30, 109–113 (1993).
Iserentant D., Geenens W., Verachtert H.: Titrated acidification power: a simple and sensitive method to measure yeast vitality and its relation to other vitality measurements.J.Am.Soc.Brew.Chem. 54, 110–114 (1996).
Kara B.V., Simpson W.M., Hammond R.M.: Prediction of the fermentation performance of brewing yeast with the acidification power test.J.Inst.Brew. 94, 153–158 (1988).
Lawrence D.R.: Brewing yeast fermentation performance.Internat.J.Food Sci.Technol. 37, 337–338 (2002).
Malfeito-Ferreira M., Guerra J.P.M., Loureiro V.: Proton extrusion as indicator of the adaptive state of yeast starters for the continuous production of sparkling wines.Am.J.Enol.Viticult. 41, 219–222 (1990).
Mathieu C., Van den Bergh L., Isenrentant D.: Prediction of yeast fermentation performance using the acidification power test. pp. 273–280 inProc. 23rd Internat. Congr. Eur. Brew. Convention, Lisbon (Portugal) 1991.
Opekarová M., Sigler K.: Acidification power: indicator of metabolic activity and autolytic changes inSaccharomyces cerevisiae.Folia Microbiol. 27, 395–403 (1982).
Patino H., Edelen C., Miller J.: Alternative measures of yeast vitality: use of cumulative acidifcation power and conductance.J.Am.Soc.Brew.Chem. 51, 128–132 (1993).
Peddie F.L., Simpson W.J., Kara B.V., Robertson S.C., Hammond J.R.M.: Measurement of endogenous oxygen uptake rates of brewer’s yeast.J.Inst.Brew. 97, 21–25 (1991).
Quain D.E.: Studies on yeast physiology — impact on fermentation performance and product quality.J.Inst.Brew. 89, 38–40 (1988).
Rechinger K.B., Siegumfeldt H.: Rapid assessment of cell viability ofLactobacillus delbrueckii subsp.bulgaricus by measurement of intracellular pH in individual cells using fluorescence ratio imaging microscopy.Internat.J.Food Microbiol. 75, 53–60 (2002).
Riis S.B., Pedersen H.M., Sorensen N.K., Jakobsen M.: Flow cytometry and acidification power test as rapid techniques for determination of the activity of starter cultures ofLactobacillus delbrueckii spp.bulgaricus.Food Microbiol. 12, 245–250 (1995).
Rodrigues P.G., Barros A.A., Rodrigues J.A., Ferreira A.A., Goncalves C., Hammond J.R.M.: Yeast quality control in industrial brewing process using vitaltitration, a new method for vitality determination. Contribution 57,Proc. 29th Internat. Congr. Eur. Brew. Convention, Dublin (Ireland) 2003.
Semjonovs P., Marauska M., Linde R., Grube M., Zikmanis P., Bekers M.: Development ofBifidobacterium lactis Bb 12 on β-(2,6)-linked fructan-containing substrate.Eng.Life Sci. 4, 433–437 (2004).
Shen H.Y., Moonjai N., Verstrepen N., Delvaux F.R.: Impact of attachment immobilization on yeast physiology and fermentation performance.J.Am.Soc.Brew.Chem. 61, 79–87 (2003).
Sigler K., Knotková A., Kotyk A.: Factors governing substrate-induced generation and extrusion of protons in the yeastSaccharomyces cerevisiae.Biochim.Biophys.Acta 643, 572–582 (1981a).
Sigler K., Kotyk A., Knotková A., Opekarová M.: Processes involved in the creation of buffering capacity and in substrate-induced proton extrusion in the yeastSaccharomyces cerevisiae.Biochim.Biophys.Acta 643, 583–592 (1981b).
Sigler K., Pascual C., Romay C.: Intracellular control of proton extrusion inSaccharomyces cerevisiae.Folia Microbiol. 28, 363–370 (1983).
Šusta J., Hodaň J., Opekarová M., Sigler K.: A simple method for determining the metabolic activity of brewer’s yeast during the brewing process.Food Microbiol. 1, 169–171 (1984).
Van Zandycke S.M., Siddique R., Smart K.A.: Yeast membrane potential and carbohydrate utilization, pp. 334–343 inProc. 28th Internat. Congr.Eur.Brew. Convention, Budapest (Hungary) 2001.
Vincent S.F., Bell P.J.L., Bissinger P., Nevalainen K.M.-H.: Comparison of melibiose utilizing baker’s yeast strains produced by genetic engineering and classical breeding.Lett.Appl.Microbiol. 28, 148–152 (1999).
Wackerbauer K., Ludwig A., Mohle J.: Improvement of long term stability in main fermentations with immobilized yeast — II. Trials in MPI-reactor and evaluation of yeast vitality in the immobilized system.Monatsschr.Brauwissen. 55, 172–181 (2002).
Yoshikawa S., Chikara K.I., Hashomoto H., Mirsui N., Shimosaka M., Okazaki M.: Isolation and characterization ofZygosaccharomyces rouxii mutants defective in proton pump out activity and salt tolerance.J.Ferment.Bioeng. 79, 6–10 (1995).
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by Research Center IM 0570 of theMinistry of Education, Youth and Sports of the Czech Republic and by theInstitutional Research Concepts of theInstitute of Microbiology (AV 0Z 5020 0510), theResearch Institute of Brewing and Malting (MSM 601 936 9701) andFaculty of Mathematics and Physics, Charles University (MSM 002 162 0835).
Rights and permissions
About this article
Cite this article
Sigler, K., Mikyška, A., Kosař, K. et al. Factors affecting the outcome of the acidification power test of yeast quality: Critical reappraisal. Folia Microbiol 51, 525–534 (2006). https://doi.org/10.1007/BF02931616
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02931616