Abstract
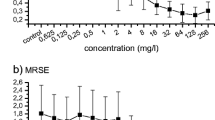
Due to high resistance, standard chemotherapy of biofilm-associated staphylococcal infections is ineffective and a number of alternative approaches to antimicrobial treatment have been proposed. Minimum inhibitory concentration (MIC) and biofilm inhibitory concentration (BIC) of oxacillin (Oxa), vancomycin (Van), linezolid (Lzd) and lysostaphin (Lss) as well as the possible synergistic effect of the antibiotics and lysostaphin were determined. The Lss susceptibility ofStaphylococcus aureus planktonic and biofilm cultures varied and was strain-dependent. The synergistic effect of sub-BICLss+Oxa was observed for methicillin-sensitiveS. aureus (Ms Sa) and methicillin-resistantS. aureus (Mr Sa), but not for heterogeneously vancomycin-resistantS. aureus (Vh Sa) biofilm. Van with sub-BICLss was effective against Ms Sa and Mr Sa biofilm, when applied in three subsequent doses. Only sub-BICLss+Lzd combination, given as three cycles therapy, was effective in disruption of all 3 (Ms Sa, Mr Sa, Vh Sa) biofilms.
Similar content being viewed by others
Abbreviations
- BIC:
-
biofilm inhibitory concentration
- Mr Sa :
-
methicillin-resistantS. aureus
- FITC:
-
fluorescein isothiocyanate
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- Vh Sa :
-
S. aureus heterogeneously resistant to vancomycin
- Lss:
-
lysostaphin
- MIC:
-
minimal inhibitory concentration
- Ms Sa :
-
methicillin-sensitiveS. aureus
- Lzd:
-
linezolid
- Oxa:
-
oxacillin
- Van:
-
vancomycin
References
Barequet I.S., Ben Simon G.J., Safrin M., Ohman D.E., Kessler E.:Pseudomonas aeruginosa LasA protease in treatment of experimental staphylococcal keratitis.Antimicrob.Agents Chemother. 48, 1681–1687 (2004).
Boyle-Vavra S., Carey R.B., Daum R.S.: Development of vancomycin and lysostaphin resistance in a methicillin-resistantStaphylococcus aureus isolate.J.Antimicrob.Chemother. 48, 617–625 (2001).
Černohorská L., Votava M.: Determination of minimal regrowth concentration (MRC) in clinical isolates of various biofilm-forming bacteria.Folia Microbiol. 49, 75–78 (2004).
Climo M.W., Ehlert K., Archer G.L.: Mechanism and suppression of lysostaphin resistance in oxacillin-resistantStaphylococcus aureus.Antimicrob.Agents Chemother. 45, 1431–1437 (2001).
Dajcs J.J., Thibodeaux B.A., Girgis D.O., Shaffer M.D., Delvisco S.M., O’Callaghan R.J.: Immunity to lysostaphin and its therapeutic value for ocular MRSA infections in the rabbit.Invest.Ophthalmol.Visual Sci. 43, 3712–3716 (2002).
Drago L., Vecchi E.D., Valli M., Nicola L., Gismondo M.R.: Effect of linezolid in comparison with that of vancomycin on glycocalyx production:in vitro study.Antimicrob.Agents Chemother. 46, 598–599 (2002).
El-Azizi M., Rao S., Kanchanapoom T., Khardori N.:In vitro activity of vancomycin, quinupristin/dalfopristin, and linezolid against intact and disrupted biofilms of staphylococci.Ann.Clin.Microbiol.Antimicrob. 4, 1–9 (2005).
Fux C.A., Stoodley P., Hall-Stoodley L., Costerton J.W.: Bacterial biofilms: a diagnostic and therapeutic challenge.Expert Rev. Anti-infect.Ther. 1, 667–683 (2003).
Gander S., Hayward K., Finch R.: An investigation of the antimicrobial effects of linezolid on bacterial biofilms utilizing anin vitro pharmacokinetic model.J.Antimicrob.Chemother. 49, 301–308 (2002).
Goetz F.:Staphylococcus and biofilms.Mol.Microbiol. 43, 1367–1378 (2002).
Hall-Stoodley L., Costerton J.W., Stoodley P.: Bacterial biofilms: from the natural environment to infectious diseases.Nature Rev. 2, 95–106 (2004).
Kairo S.K., Bedwell J., Tyler P.C., Carter A., Corbel M.J.: Development of a tetrazolium salt assay for rapid determination of viability of BCG vaccines.Vaccine 17, 2423–2428 (1999).
Kiri N., Archer G., Climo M.W.: Combinations of lysostaphin with β-lactams are synergistic against oxacillin-resistantStaphylococcus epidermidis.Antimicrob.Agents Chemother. 46, 2017–2020 (2002).
Lewis K.: Riddle of biofilm resistance.Antimicrob.Agents Chemother. 45, 999–1007 (2001).
National Committee for Clinical Laboratory Standards: Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically.Approved Standard 5th ed. M7-A5, 1–54 (2000).
Pitts B., Hamilton M.A., Zelver N., Stewart P.S.: A microtiter-plate screening method for biofilm disinfection and removalJ.Microbiol.Meth. 54, 269–276 (2003).
Prakash B., Veeregowda B.M., Krishnappa G.: Biofilms: a survival strategy of bacteria.Curr.Sci. 85, 1299–1307 (2003).
Růžička F., Hola V., Votava M., Tejkalová R., Horvát R., Heroldova M., Woznicova V.: Biofilm detection and the clinical significance ofStaphylococcus epidermidis isolates.Folia Microbiol. 49, 75–78 (2004).
Walencka E., Sadowska B., Różalska S., Hryniewicz W., Różalska B.: Lysostaphin as a potential therapeutic agent for staphylococcal biofilm eradication.Polish J.Microbiol. 54, 191–200 (2005).
Wilcox H.M.: Efficacy of linezolidversus comparator therapies in Gram-positive infections.J.Antimicrob.Ther. 51 (Suppl. S2), ii27-ii35 (2003).
Wu J.A., Kusuma C., Mond J.J., Kokai-Kun J.F.: Lysostaphin disruptsStaphylococcus aureus andStaphylococcus epidermidis biofilms on artificial surfaces.Antimicrob.Agents Chemother. 47, 3407–3414 (2003).
Author information
Authors and Affiliations
Additional information
This study was supported by the grant 2PO5A 149 29 from thePolish Scientific Research Committee.
Rights and permissions
About this article
Cite this article
Walencka, E., Sadowska, B., Róžalska, S. et al. Staphylococcus aureus biofilm as a target for single or repeated doses of oxacillin, vancomycin, linezolid and/or lysostaphin. Folia Microbiol 51, 381–386 (2006). https://doi.org/10.1007/BF02931580
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02931580