Summary
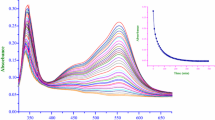
Solubilities of salts of iron(II) complexes of tridentate imine (Schiff base) ligands are reported for aqueous solution and for solutions in methanol-water mixtures. Gibbs free energies of transfer for the iron(II) complexes have been calculated, using the TPTB assumption (i.e. assuming equality of transfer parameters for Ph4P+ and Ph4B−). The derived parameters demonstrate the importance both of ion size and of hydrophilic/hydrophobic character in determining ionic solvation in binary aqueous mixtures. Kinetic data are reported for reaction of these iron(II) complexes with hydroxide in methanol—water mixtures. The transfer parameters derived for the complexes are used in the analysis of the kinetic data to describe the influence of solvent composition on the initial and transition states. Differences between this TPTB-based analysis and the results of a similar analysis based on Wells's solvent-sorting approach are discussed.
Similar content being viewed by others
References
T. S. Lee, I. M. Kolthoff and D. L. Leussing,J. Am. Chem. Soc.,70, 3596 (1948); D. W. Margerum and L. P. Morgenthaler,J. Am. Chem. Soc.,84, 706 (1962); M. J. Blandamer, J. Burgess and D. L. Roberts,J. Chem. Soc., Dalton Trans., 1086 (1978).
J. Burgess,J. Chem. Soc. A, 955 (1967); 497 (1968).
M. J. Blandamer, J. Burgess, R. I. Haines, F. M. Mekhail, and P. Askalani,J. Chem. Soc., Dalton Trans., 1001 (1978).
R. D. Gillard, D. W. Knight and P. A. Williams,Transition Met. Chem.,5, 321 (1980).
R. D. Gillard,Inorg. Chim. Acta,11, L21 (1974); R. D. Gillard,Coord. Chem. Rev.,50 303 (1983); N. Serpone, G. Ponterini, M. A. Jamieson, F. Bolletta and M. Maestri,Coord. Chem. Rev.,50, 209 (1983); E. C. Constable,Polyhedron,2, 551 (1983).
P. Krumholz,Inorg. Chem.,4, 612 (1965).
F. P. Dwyer, N. S. Gill, E. C. Gyarfas and F. Lions,J. Am. Chem. Soc.,75, 3834 (1953).
R. K. Murmann and E. A. Healy,J. Am. Chem. Soc.,83, 2092 (1961).
H. A. Goodwin and F. Lions,J. Am. Chem. Soc.,82, 5013 (1960).
J. Burgess and R. H. Prince,J. Chem. Soc., 6061 (1965); R. Farina, R. Hogg and R. G. Wilkins,Inorg. Chem.,7, 170 (1980); J. Burgess and M. V. Twigg,J. Chem. Soc., Dalton Trans., 2032 (1974).
R. D. Gillard,Coord. Chem. Rev.,16, 67 (1975); R. D. Gillard C. T. Hughes and P. A. Williams,Transition Met. Chem.,1, 51 (1976).
J. Burgess and C. D. Hubbard,Comments Inorg. Chem., to be submitted.
M. J. Blandamer and J. Burgess,Coord. Chem. Rev.,31, 93 (1980).
M. J. Blandamer, J. Burgess, B. Clark, P. P. Duce, A. W. Hakin, N. Gosal, S. Radulović, P. Guardado, F. Sanchez, C. D. Hubbard and E. A. Abu-Gharib,J. Chem. Soc. Faraday Trans. I,82, 1471 (1986).
M. J. Blandamer, J. Burgess and E. A. Abu-Gharib,Transition Met. Chem.,9, 193 (1984).
J. Burgess and E. A. Abu-Gharib,Transition Met. Chem.,9, 234 (1984).
E. A. Abu-Gharib, M. J. Blandamer, J. Burgess, N. Gosal, P. Guardado, F. Sanchez and C. D. Hubbard,Transition Met. Chem.,9, 306 (1984).
C. Tissier,Comptes Rend.,286C, 35 (1978).
M. H. Abraham, T. Hill, H. C. Ling, R. A. Schulz and R. A. C. Watt,J. Chem. Soc., Faraday Trans. I,80, 489 (1984).
J. Burgess and C. D. Hubbard,J. Chem. Soc., Chem. Commun., 1482 (1983).
C. F. Wells,J. Chem. Soc., Faraday Trans. I,70, 694 (1974).
J. Burgess,Mech. Inorg. Organometal. React.,1, 151 (1983).
Author information
Authors and Affiliations
Additional information
TMC 2612
Rights and permissions
About this article
Cite this article
El-Samahy, A.A., Abu-Gharib, EE.A., Eltaher, AE. et al. Solvent effects on reactivity trends for base hydrolysis of iron(II) complexes of tridentate Schiff base imine ligands. Transition Met. Chem. 17, 438–442 (1992). https://doi.org/10.1007/BF02910727
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02910727