Abstract
Objective
Cadmium is known to affect the vascular tone of isolated blood vesselsin vitro and the arterial pressure of ratsin vivo. However, the mechanisms of cadmium actions on the vascular system have not been clarified. To elucidate the actions of cadmium on vascular tonus, effects of cadmium on vasocontractile and vasorelaxant responsesin vitro were investigated using aortic strips isolated from rats.
Methods
Aortic strips isolated from male Wistar rats were incubated with CdCl2 (10μM) for 24 hr, washed with fresh CdCl2-free medium, and then used for measurement of isometric tension and Western blot analysis of eNOS (endothelial nitric oxide synthase) and iNOS (inducible nitric oxide synthase).
Results
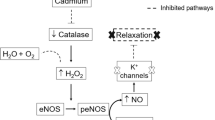
In the aortas pretreated with cadmiumin vitro, the contractile response to phenylephrine was significantly higher than that in the control aortic strips pretreated with a vehicle. The sodium nitroprusside-induced relaxing response was significantly higher in the aortic strips pretreated with cadmium for 24 hr, compared with that in the control pretreated with a vehicle. The isoproterenol-induced relaxing response was also significantly higher in the cadmium-accumulated aortic strips.In vitro cadmium treatment slightly but not significantly increased the acetylcholine-induced relaxation of the aortic strips. Cadmium treatment induced expression of iNOS and significantly increased expression of eNOS in the aortic strips, while it did not affect expression of β-actin.
Conclusions
Cadmium treatmentin vitro augmented the α1 adrenoceptor-mediated contractile response, even though eNOS and iNOS were upregulated by cadmium treatment. NO-induced and β-adrenoceptor-mediated relaxing responses were also augmented by cadmium treatment. These results suggest that both vasocontractile and vasorelaxing responses are augmented in cadmium-accumulated aortas.
Similar content being viewed by others
References
Perry HM, Schroeder HA. Concentration of trace metals in urine of treated and untreated hypertensive patients compared with normal subjects. J. Lab. Clin. Med. 1955;46:936.
Glauser SC, Bello CT, Glauser EM. Blood-cadmium levels in normotensive and untreated hypertensive humans. Lancet 1976;1:717–718.
Medeiros DM, Pellum LK. Elevation of cadmium, lead, and zinc in the hair of adult black female hypertensives Bull. Environ. Contam. Toxicol. 1984;32:525–532.
Kagamimori S, Naruse Y, Fujita T, Watanabe M, Nishino H, Shinmura T. Factors associated with blood pressure in females with heavy exposure to cadmium. Bull. Environ. Contam. Toxicol. 1985;35:386–392.
Schroeder HA, Vinton Jr WH. Hypertension induced in rats by small doses of cadmium. Am. J. Physiol. 1962;202:515–518.
Perry Jr HM, Erlanger M, Perry EF. Elevated systolic pressure following chronic low-level cadmium feeding. Am. J. Physiol. 1977;232:H114-H121.
Perry Jr HM, Erlanger MW. Metal-induced hypertension following chronic feeding of low doses of cadmium and mercury. J. Lab. Clin. Med. 1974;83:541–547.
Eakin DJ, Schroeder LA, Whanger PD, Weswig PH. Cadmium and nickel influence on blood pressure, plasma renin, and tissue mineral concentrations. Am. J. Physiol. 1980;238:E53-E61.
Revis N. A possible mechanism for cadmium-induced hypertension in rats. Life Sci. 1978;22:479–487.
Williams BJ, Laubach DJ, Nechay BR, Steinsland OS. The effect of cadmium on adrenergic neurotransmission in vitro. Life Sci. 1978;23:1929–1933.
Aoshima K, Kasuya M. Environmental exposure to cadmium and effects on human health. Part 3. The results of blood examinations and blood pressure in inhabitants of the cadmium-polluted Jinzu river basin in Toyama Prefecture. Jpn. J. Hyg. 1988;43:949–955.
Perry Jr HM, Schoepfle E, Bourgoignie J. In vitro production and inhibition of aortic vasoconstriction by mercuric, cadmium, and other metal ions. Proc. Soc. Exp. Biol. Med. 1967; 124:485–490.
Thind GS, Stephan KF, Blakemore WS. Inhibition of vasopressor responses by cadmium. Am. J. Physiol. 1970;219:577–583.
Toda N. Influence of cadmium ions on contractile response of isolated aortas to stimulatory agents. Am. J. Physiol. 1973; 225:350–355.
Niwa A, Suzuki A. Effects of cadmium on the tension of isolated rat aorta (a possible mechanism for cadmium-induced hypertension). J. Toxicol. Sci. 1982;7:51–60.
Balaraman R, Rathod SP, Gulati OD. Effect of cadmium on contractile response to spasmogens in vascular and nonvascular tissues. Indian J. Exp. Biol. 1990;28:455–459.
Zhang C, Thind GS, Joshua IG, Fleming JT. Cadmium-induced arteriolar constriction in skeletal muscle microcirculation. Am. J. Hypertens. 1993;6:325–329.
Skoczynska A, Wrobel J, Andrzejak R. Lead-cadmium interaction effect on the responsiveness of rat mesenteric vessels to norepinephrine and angiotensin II. Toxicology 2001;162:157–170.
Ozdem SS, Ogutman C. The effects of short-term nifedipine treatment on responsiveness of aortic rings of cadmium-hypertensive rats. Clin. Exp. Hypertens. 1999;21:423–440.
Furchgott RF. Role of endothelium in responses of vascular smooth muscle. Circ. Res. 1983;53:557–573.
Toda N, Okamura T. Different susceptibility of vasodilator nerve, endothelium and smooth muscle functions to Ca++ antagonists in cerebral arteries. J. Pharmacol. Exp. Ther. 1992;261:234–239.
MacLeod KM, Ng DD, Harris KH, Diamond J. Evidence that cGMP is the mediator of endothelium-dependent inhibition of contractile responses of rat arteries to α-adrenoceptor stimulation. Mol. Pharmacol. 1987;32:59–64.
Bolton TB, MacKenzie I, Aaronson PI. Voltage-dependent calcium channels in smooth muscle cells. J. Cardiovasc. Pharmacol. 1988;12 Suppl 6:S3–7.
Wakabayashi I, Sakamoto K, Hatake K. Inhibitory effects of cadmium ion on extracellular Ca2+-independent contraction of rat aorta. Eur. J. Pharmacol. 1995;293:133–140.
Abu-Hayyeh S, Sian M, Jones KG, Manuel A, Powell JT. Cadmium accumulation in aortas of smokers. Arterioscler. Thromb. Vasc. Biol. 2001;21:863–867.
Chiu AT, Bozarth JM, Timmermans PB. Relationship between phosphatidylinositol turnover and Ca2+ mobilization induced by α-1 adrenoceptor stimulation in the rat aorta. J. Pharmacol. Exp. Ther. 1987;240:123–127.
Sakurada K, Wakabayashi I. Cadmium accumulation augments contraction and phosphoinositide hydrolysis of vascular smooth muscles. Res. Commun. Mol. Pathol. Pharmacol. 1999;106:212–220.
Shepherd JT. Increased systemic vascular resistance and primary hypertension: the expanding complexity. J. Hypertens. 1990;8(Suppl 7):S15-S27.
Ignarro LJ. Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ. Res. 1989;65:1–21.
Kaji T, Mishima A, Yamamoto C, Sakamoto M, Koizumi F. Effect of cadmium on the monolayer maintenance of vascular endothelial cells in culture. Toxicology 1992;71:267–276.
Kishimoto T, Oguri T, Ohno M, Matsubara K, Yamamoto K, Tada M. Effect of cadmium (CdCl2) on cell proliferation and production of EDRF (endothelium-derived relaxing factor) by cultured human umbilical arterial endothelial cells. Arch Toxicol 1994;68:555–559.
Rees DD, Palmer RM, Schulz R, Hodson HF, Moncada S. Characterization of three inhibitors of endothelial nitric oxide synthase in vitro and in vivo. Br. J. Pharmacol. 1990;101:746–752.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takahashi, Y., Poteser, M., Masui, H. et al. Effects of cadmiumin vitro on contractile and relaxant responses of isolated rat aortas. Environ Health Prev Med 9, 251–256 (2004). https://doi.org/10.1007/BF02898138
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02898138