Abstract
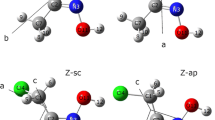
The conformational analyses of α-mono, α-di and α-trichloroacetanilides have been carried out using IR spectroscopy. α-Monochloro, dichloro and trichloroacetanilides were found to exist in only one conformation in CH2Cl2 and CHC13. The existence of all these anilides in single conformation is due to intramolecular H-bond formation between N−H and Cl.
In CH3CN, the broadening of C–0 bands of monochloroacetanilides and their bifurcation in α-dichloroacetanilides indicated the existence of two rotamers due to the breaking of chelation by the solvent.
Similar content being viewed by others
References
Bellamy L J and Williams R L 1957J. Chem. Soc. 4294
Bellamy L J and Williams R L 1958J. Chem. Soc. 3465
Brown T L 1958J. Am. Chem. Soc. 80 3513
Letaw H and Gropp A H 1953J. Chem. Phys. 21 1621
Mizushima S, Shimanouch T, Ichishima I, Miyazawa T, Nakagawa I and Araki T 1956J. Am. Chem. Soc. 78 2038
Nyquist R A 1963aSpectrochim. Acta 19 509
Nyquist R A 1963bSpectrochim. Acta 19 1595
Suryanarayana I, Subrahmanyam B and Subba Rao N V 1975Proc. Indian Acad. Sci. A82 55
Vogel A I 1980Text-book of practical organic chemistry (4th edn) (London: ELBS and Longmans) p. 684
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Venkateshwarlu, G., Reddy, P.P. & Subrahmanyam, B. Conformations ofαchloroacetanilides: An IR spectroscopic study. Proc. Indian Acad. Sci. (Chem. Sci.) 101, 75–82 (1989). https://doi.org/10.1007/BF02869388
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02869388