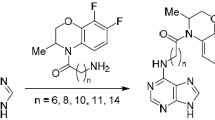
Abstract
The nucleophilic substitution reaction of 6-chloro purines (I) with malononitrile and ethyl cyanoacetate is carried out in DMSO and in the presence of an alkali. The possible tautomeric-ylidene form for the products is considered and discussed in view of IR, UV, NMR and mass spectral determinations. The derivatives were tested for their antitumor activities.
Similar content being viewed by others
Literature Cited
Glazer: Analysis of the effect of 5-F.U on the synthesis and translation of polysomal poly (A) RNA from Ehrlich ascites cells,Cancer Chemother. Pharmacol. 4, 227 (1980).
Flersher, M.H.: Some short-chain N6-substituted adenosine analogues with antitumor properties,J. Med. Chem. 23, 1448 (1980).
Bloch, A.: Synthesis and crystal structure of 4-(2-deoxy-β-D-erythro-pentofuranosyl)-6-methyl-1,2,4-triazin-3(H)-one-1-oxide, a structural analogy o thymidie,New Leads Cancer Ther. 1, 65 (1981).
Moharram, H.H.: Reaction of purine derivatives with phosphorus pentaoxide and triethylamine and their antiumor effects,Arch. Pharm. Res. 12, 1 (1988).
Moharram, H.H.: Reactions with purine derivatives: synthesis of arylamino purines of antitumor effects,Arch. Pharm. Res. (1988) (submitted).
Mizuno, Y.: Extension of malonic ester synthesis to the heterocyclic series,Chem. Pharm. Bull. (Tokyo),2, 225 (1954).
Hayashi, E.: Purines II, on 6-(methylsulfonyl)-9-phenyl-9H-purine,J. Pharm. Soc. (Japan),99, 201 (1979).
El-Bayouki, K.A.M.: Purine derivatives for the regulation of plant growth,Eur. Patent. Appl., Ep 155,911;Chem. Abstr. 104, 186245C (1986).
Hayashi, E.: Purines III, Reaction of 6-chloro-9-phenyl-9H-purine with carbanion,J. Pharm. Soc. (Japan) 99, 205 (1979).
Bellamy, L.J.: IR spectra of complex molecules, John wiley and sons, Inc. New York, N.Y. p. 255 (1988).
Mason, S.F.: Purine studies, part II. The Ultra-violet absorption spectra of some mono-and polysubstituted purines,J. Chem. Soc. 2071 (1954).
Matsuura, S.: Heterocyclic chemistry. part II. Nuclear magnetic resonance studies of purines and pteridines,J. Chem. Soc. 623 (1965).
El-Bayouki, K.A.M.: Ethylchloroformate (DMF) in organic synthesis: (I) A novel reagent for ring closure of 1-aryl-5-amino-4-imidazolecarboxamides to their hypoxanthine derivatives,Gazz-chem. Ital. accepted for publication (in press) (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moharram, H.H., El-Bayouki, K.A.M., Haggag, B. et al. Synthesis and structure of purine derivatives as antitumor effects. Arch. Pharm. Res. 12, 138–142 (1989). https://doi.org/10.1007/BF02857738
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02857738