Abstract
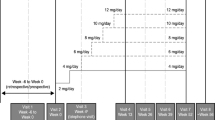
This 6-month open-label study evaluated the efficacy and safety of topiramate monotherapy for partial-onset or primarily generalized seizures in 22 adults with newly diagnosed epilepsy. The initial topiramate dose of 25 mg could be titrated to a maximum effective or maximum tolerated dose of 200 mg/d. Eighteen patients completed the study. The mean topiramate dose was 200 mg/d (range, 75–200 mg/d). Compared with baseline, monthly seizure frequency decreased 89% (median), with 17 patients (94%) achieving complete control. Five patients dropped out of the study. No changes in hematologic, renal, and hepatic function evaluations were noted, and only 1 patient experienced clinical signs significant enough to warrant discontinuation of treatment. In this small study, topiramate in doses ranging from 75 to 200 mg/d was effective and well tolerated as monotherapy in adults with newly diagnosed epilepsy.
Similar content being viewed by others
References
Brodie MJ, Dichter MA. Antiepileptic drugs.N Engl J Med. 1996;334:169–175.
Sachdeo RC, Reife RA, Lim P, Pledger G. Topiramate monotherapy for partial onset seizures.Epilepsia. 1997;38:294–300.
Gilliam FG, Reife R, Wu S-C. Topiramate monotherapy: randomized controlled trial in patients with recently diagnosed localization-related epilepsy.Neurology. 1999;52(suppl 2):A248.
Privitera MD, Brodie MJ, Neto W, Wang S. Topiramate vs investigator choice of carbamazepine or valproate as monotherapy in newly diagnosed epilepsy.Epilepsia. 2000;41(suppl 7):93–94.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Salinas-Estabane, R. Topiramate monotherapy in adults with newly diagnosed epilepsy. Adv Therapy 19, 126–128 (2002). https://doi.org/10.1007/BF02850268
Issue Date:
DOI: https://doi.org/10.1007/BF02850268