Abstract
The injectable third-generation cephalosporin, cefoperazone, was introduced in India in early 1994. A post-marketing surveillance study was conducted in 1994–95 to collect local data on efficacy and safety, evaluate the native pattern of bacterial susceptibility, and assess the relationship ofin vitro susceptibility tests to clinical efficacy. Hospitalized patients with features suggestive of infections that are approved indications for cefoperazone were eligible for the study. This report presents an analysis of the results among patients in the pediatric age group. The recommended dose range for children was 50–200 mg/kg/day. At analysis, 95 patients aged 6 days to 14 years were evaluable for efficacy. About a third of these (31%) were judged to have hospital acquired infections, and 22 (23%) had failed prophylaxis or previous treatment.
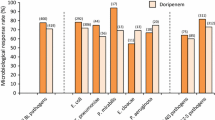
A successful clinical outcome was observed in 91% of 75 non-neutropenic patients. Eighteen (95%) of 19 patients with pneumonia and 8 (89%) of 9 patients with pyogenic meningitis responded to cefoperazone. Five of 6 patients with complicated or hospital-acquired upper urinary tract infections, 2 of 3 patients with peritonitis, and all patients with skin or soft tissue infections, intra-abdominal abscess, ostemyelitis, and brain abscess also responded. Four of 5 non-neutropenic patients with septicemia recovered with cefoperazone monotherapy. The response rate was 70% among 20 neutropenic patients, 16 of whom were deemed septicemic. Antibiotic disc susceptibility data among pediatric patients was available for 63 isolates and 13% were reported resistant to cefoperazone. Microbiologic eradication was reported with 94% of initial isolates. Mild to moderate adverse events of study, drug-related or uncertain causation occurred in 4 (3.7%) of patients.
Similar content being viewed by others
References
Gordon AJ, Phyfferoen M. Cefoperazone sodium in the treatment of serious infections in 2,100 adults and children; multicentered trials in Europe, Latin America, and Australia.Rev Infect Dis 1983; 5 (Suppl): S188-S199.
Barillari A, Devriendt E, Cieters P,et al. Open multicenter study of cefoperazone in the treatment of severe infections in the pediatric age group.Curr Ther Res 1987; 41: 57–64.
Lee JS, Kim DH, Chin DS,et al. Clinical study of cefoperazone in the treatment of bacterial infection in the practice of pediatrics.New Med J (Korea), 1985; 28: 65–76.
Kobayashi Y, Morikawa Y, Haruta T, Fujiwara T. Clinical evaluation of cefoperazone in children.Jpn J Antibiot 1980; 33: 910–915.
Matsuda H, Yoshida T, Kagata N, Niino S. Clinical examination of cefoperazone in pediatrics.Jpn J Antibiot 1980; 33: 935–940.
Motohiro T, Sakata Y; Fujimoto T;et al. Treatment of whooping cough and whooping cough syndrome by cefoperazone (author’s trans).Jpn J Antibiot 1980; 33: 1183–1193.
Pape JW, Gerdes H, Orioll. Typhoid fever; Successful therapy with cefoperazone.J Infect Dis 1986; 153: 272–276.
Soe GB, Overturf GD. Treatment of typhoid fever and other systemic salmonelloses with cefotaxime, ceftriaxone, cefoperazone, and other newer cephalosporins.Rev Infect Dis 1987; 9: 719–736.
Neu HC. A review and summary of the pharmacokinetics of cefoperazone; a new, extended-spectrum β-lactam antibiotic.Ther Drug Monitoring 1981; 3: 121–128.
Chandrasekar PH. Bacterial infections in hospitalized patients.Ind J Int Med 1995; 5 (suppl 5): 3–11.
Saito A, Ueda Yasushi. Multicenter clinical trials of cefoperazone in Japan.Clinical Therapeutics 1984; 7 (1): 49–59.
The cefoperazone collaborative post-marketing surveillance study group. Cefoperazone: Results of a post-marketing surveillance study in patients and correlation ofin vitro susceptibility tests with clinical outcome.Hosp Today 1997; 2(5): 21–30.
Cristiano P, Pempinello R, Iovene MR, Coppola L, Altucci P. Cefoperazone therapy of bacterial meningits; a clinical trial.Infection 1989; 17 (6): 378–81.
Kitamura K, Shimizu T, Abe H,et al. Clinical studies of cefoperazone in neurosurgery.Jpn J Antibiot 1986; 39 (1): 63–74.
Ueda Y. Clinical and bacteriological efficacy of cefoperazone sodium in world-wide studies of 4,485 adults and paediatric patients.International Medicine 1983; 2 (3): 15–20.
Murray PR. Antimicrobial susceptibility tests; Testing methods and interpretative problems.In: Poupard JA, Walsh AR, Kleger B, eds. Antimicrobial susceptibility testing: Critical issues for the 90s.Advances in Experimental Medicine and Biology, Vol 349, New York: Plenum Press, 1994; 15–25.
Author information
Authors and Affiliations
Consortia
Rights and permissions
About this article
Cite this article
The Cefoperazone Collaborative Post-marketing Surveillance Study Group., Mukherjee, S. Cefoperazone: An analysis of results in the pediatric population from a post-marketing surveillance study in hospitalized patients. Indian J Pediatr 65, 89–98 (1998). https://doi.org/10.1007/BF02849699
Issue Date:
DOI: https://doi.org/10.1007/BF02849699