Abstract
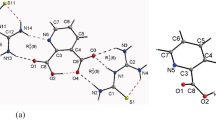
The IR and Raman spectra are measured and analysed for sodium pyrophosphate decahydrate. The spectra are interpreted on the basis of P2O 4−7 ion and water vibrations. The observed results fit with the features predicted for the factor goup model. The appearance of two sets of frequencies in the stretching and bending regions of water suggests the existence of two kinds of water molecules in the crystal. This is confirmed by deuterium substitution.
Similar content being viewed by others
References
Calvo C 1909Acta Crystallogr. 23 289
Cornilsen B C and Condrate R A 1977J. Phys. Chem. Solids 38 1327
Cornilsen B C and Condrate R A 1978J. Solid State Chem. 23 375
Hanwick T I and Hoffmann P O 1951J. Chem. Phys. 19 708
Hezel A and Ros S D 1967Spectrochim. Acta A23 1583
Mac Arthur D M and Beevars C A 1957Acta Crystallogr. 10 428
Palmer W G 1961J. Chem. Soc. 1552
Saksena B D 1961Trans. Faraday Soc. 57 242
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ramakrishnan, V., Kalkura, S.N. & Rajagopal, P. Vibrational spectra of Na4P2O7·10H2O. Pramana - J Phys 34, 555–560 (1990). https://doi.org/10.1007/BF02846431
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02846431