Abstract
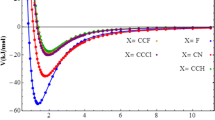
Ab initio supermolecular SCF calculations at the STO-3G level are reported for the diacetylene-hydrogen fluoride complexes. The reverse σ-complex is predicted to have somewhat higher stability and H-bond strength than the π-complexes.
Similar content being viewed by others
References
Andrews L, Johnson G L and Kelsall B J 1982aJ. Chem. Phys. 76 5767
Andrews L, Johnson G L and Kelsall B J 1982bJ. Chem. Phys. 86 3374
Andrews L. Johnson G L and Davis S R 1985J. Chem. Phys. 89 1706
Barnes A J, Davies J B, Hallam H E and Howells J D R 1973J. Chem. Soc., Faraday Trans. 2 69 246
Baiocchi F A, Williams J W and Klemperer W 1983J. Phys. Chem. 87 2079
Legon A C, Aldrich P O and Flygare W H 1981J. Chem. Phys. 75 625
McDonald S A, Johnson G L, Keelan B W and Andrews L 1980J. Am. Chem. Soc. 102 2892
Patten K O and Andrews L 1985J. Am. Chem. Soc. 107 5594
Patten K O and Andrews L 1986J. Phys. Chem. 90 3910
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Medhi, C., Bhattacharyya, S.P. Theoretical studies on hydrogen bonding inπ-electron systems: A note on the structural features of diacetylene-hydrogen fluoride complexes. Proc. Indian Acad. Sci. (Chem. Sci.) 100, 293–295 (1988). https://doi.org/10.1007/BF02840538
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02840538