Abstract
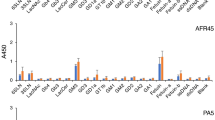
A scale of relative affinities of a series of 2′-deoxycytidine and cytidine (CD) derivatives was established based on the data of cross-reactivities of these compounds as well as the displacements obtained from a competitive ELISA. No correlation could be established between the nucleosides modifying structures and the affinities. This can be explained by the possibilities of the modifying structures of intra- and intermolecular nonimmunospecific interactions owing to their degree of functionalization.
Similar content being viewed by others
References
Arevalo J. H., Taussing M. J., and Wilson I. A. (1993) Molecular basis of cross-reactivity and the limits of antibody-antigen complementarity.Nature 365, 859–863.
Miller J. J. and Valdés R. (1992) Methods for calculating crossreactivity in immunoassays.J. Clin. Immunoassay 15, 97–107.
Pratt J. J. (1978) Steroid immunoassay in clinical chemistry.Clin. Chem. 24, 1978.
Rodríguez Tanty Ch., López-Cánovas L., López Brauet A., Rivero Paredes M., Higginson Clarke D., Vélez Castro H., Riverón Rojas A. M., and Macias Cabrera A. (1995) Introduction of an immunochemical label cytidine analogue.Nucleosides and Nucleotides 14, 219–228.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Higginson-Clarke, D., García, R.G., Tanty, C.R. et al. Monoclonal antibody affinities of structurally related modified nucleosides. Molecular and Chemical Neuropathology 28, 269–277 (1996). https://doi.org/10.1007/BF02815232
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02815232