Abstract
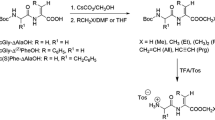
A series of fluorogenic tetra-, penta-, and hexapeptide substrates of the general structure Abz-X-Phe-Phe-Y-Ded or (-pNa in place of -Ded), where X=Ala, Ala-Ala, or Val-Ala and Y=−, Ala, or Ala-Ala, were proposed. Kinetic parameters of hydrolysis of these substrates by pepsin, cathepsin D, human gastricsin, pig pepsin, calf chymosin, and aspergillopepsin A were determined. The compounds synthesized proved to be effective substrates for aspartyl proteases of diverse origins.
Similar content being viewed by others
Abbreviations
- Abz:
-
2-aminobenzoic acid
- Ded:
-
2,4-dinitrophenylethylenediamine
- pNa:
-
p-nitroaniline
- Dnp:
-
2,4-dinitrophenyl. All amino acids areL-enantiomers
References
Kay, J. and Dunn, B.M.,Scand. J. Clin. Lab. Invest., 1992, vol. 52, pp. 23–30.
Sielecki, A.R., Fedorov, A.A., Boodhoo, A., Andreeva, N.S., and James, M.N.G.,J. Mol. Biol., 1990, vol. 214, pp. 143–170.
Blundell, T.L., Jenkins, J.A., Sewell, B.T., Pearl, L.H., Cooper, J., Tickle, I.J., Veerapandian, B., and Wood, S.P.,J. Mol. Biol., 1990, vol. 211, pp. 919–941.
Baldwin, E.T., Bhat, T.N., Gulnik, S., Hosur, M.V., Sowder R.C., II, Cachau, R.E., Collins, J., Silva, A.M., and Erickson, J.W.,Proc. Natl. Acad. Sci. USA, 1993, vol. 90, pp. 6796–6800.
Fujinaga, M., Chernaya, M.M., Tarasova, N.I., Mosimann, S.C., and James, N.G.,Protein Sci., 1995, vol. 4, pp. 960–972.
Dunn, B.N., Jimenez, M., Parten, B.F., Valler, M.J., Rolph, C.E., and Kay, J.,Biochem. J., 1986, vol. 237, pp. 899–906.
Gulnik, S.V., Suvorov, L.I., Majer, P., Collins, J., Kane, B.P., Johnson, D.G., and Erickson, J.W.,FEBS Lett., 1997, vol. 413, pp. 379–384.
Matayoshi, E.D., Wang, G.T., Krafft, G.A., and Erickson, J.W.,Science, 1990, vol. 247, pp. 954–958.
Visser, S., Slangen, C.J., and van Rooijen, P.J.,Biochem. J., 1987, vol. 244, pp. 553–558.
Dunn, B.M. and Kay, J.,Biochem. Soc. Trans., 1985, vol. 13, pp. 1041–1043.
Pohl, J. and Dunn, B.M.,Biochemistry, 1988, vol. 27, pp. 4827–4834.
Filippova, I.Yu., Lysogorskaya, E.N., Anisimova, V.V., Suvorov, L.I., Oksenoit, E.S., and Stepanov, V.M.,Anal. Biochem., 1996, vol. 234, pp. 113–118.
Zinchenko, A.A., Rumsh, L.D., and Antonov, V.K.,Bioorg. Khim., 1976, vol. 2, pp. 803–810.
Filippova, I.Yu., Lysogorskaya, E.N., Oksenoit, E.S., Komarov, Yu.E., and Stepanov, V.M.,Bioorg. Khim., 1986, vol. 12, pp. 1172–1180.
Cooper, J.B., Foundling, S.I., Blundell, T.L., Boger, J., Jupp, R.A., and Kay, J.,Biochemistry, 1989, vol. 28, pp. 8596–8603.
Fusek, M., Lin, X., and Tang, J.,J. Biol. Chem., 1990, vol. 265, pp. 1496–1501.
Kovaleva, G.G., Yusupova, M.P., Balandina, G.N., Lysogorskaya, E.N., and Stepanov, V.M.,Biokhimiya, 1977, vol. 42, pp. 534–539.
Solov’eva, T.A., Belyaev, S.V., and Stepanov, V.M.,Khim. Prir. Soedin., 1977, no. 3, pp. 398–403.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Filippova, I.Y., Lysogorskaya, E.N., Lavrenova, G.I. et al. A study of aspartyl proteases using intramolecularly quenched fluorogenic peptide substrates. Russ J Bioorg Chem 26, 169–173 (2000). https://doi.org/10.1007/BF02786342
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02786342