Abstract
Objective
To observe the pattern of changes in the proliferation and apoptosis at different stages of large bowel carcinoma in mice, and to explore the effects of the imbalance of apoptosis and proliferation at different stages of large-intestine carcinogenesis.
Methods
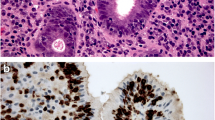
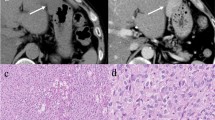
An experimental animal model for large intestine carcinogenesis of KUNMING-strain mice was used. The carcinomas were induced by subcuteneous injection of dimethylhydrazine (DMH) and the distribution and density changes of proliferating and apoptotic cells observed through multistages toward cancer formation. The animals were killed in groups at the 12th, 18th, 24th, and 32nd weeks of carcinoma induction. The apoptotic and proliferating cells were labeled separately using TUNEL and PCNA immunohistochemical staining methods
Results
In the normal mouse mucosa, all the apoptotic cells were situated in the superficial layers, however, the proliferating cells were situated in the basement layers, and the amount of both were small. In the early stage of carcinoma induction, the proliferation and the apoptotic cells slightly increased in amount, but there were no obvious changes in their ratio. In the medium stage, the densities of both distinctly increased, but there were no obvious changes in the ratio. In the late stage, the densities of the proliferating and the apoptotic cells in the non-carcinoma mucosa were higher than those at other stages. The proliferating cells in the dysplastic mucosa increased progressively with the increasing degree of the lesions. Although the apoptotic cells increased, their changes did not occur with the degree of the lesions. Their ratio showed a decreasing tendency with the degree of the lesions.
Conclusions
The presence of an imbalance between cell proliferation and apoptosis was confirmed in the course of large intestine carcinogenesis in a mouse model. 2In the early stage of carcinoma induction both proliferation and apoptosis were at a low level; in the medium stage, they were both at a high level; and in the late stage (that is in carcinoma), proliferation was at a very high level, while apoptosis was at a low level. 3The proliferating cells increased progressively with the degree of dysplasia. There were no obvious changes in the apoptotic cells and their ratio to the proliferating cells showed a progressively increasing tendency. 4ln the stage of cancer formation, the most essential change was the excessive decrease in the ratio of apoptosis to proliferation. These results support the hypothesis of “Cell Selective Proliferation”, which was raised by authors previously in a study on human large bowel carcinoma.
Similar content being viewed by others
References
Pasricha P, Bedi A, O’Conhor K, et al. The effect of sulindac on colorectal proliferation and apoptosis in familial adenomatous polyposis. Gastroenterology, 1995;109: 994–998.
Bedi A, Pastricha P, Akhtar A, et al. Inhibition of apoptosis during the development of colorectal cancer. Cancer Res, 1995;55:1811–1816.
Sun BC, Zhao XL, Hui J et al. The observation in situ of the correlation of apoptosis and proliferation in large intestinal cancer and precancerous lesions. Chin M J, 1996;76;848–852.
Arai T, Kino T. Role of apoptosis in modulation of the growth of human colorectal and villous adenomas. J Pathol, 1995;176: 37–40.
Sun BC, Zhang NX, Zhao XL, et al. The expression of cell apoptosis and the regulated gene in large bowel cancer and adenoma. Chin J Path, 1997;26: 137–140.
Reinacher-Schick A, Schoenck A, Graeven U, et al. Mesalazine causes a mitotic arrest and induces caspase-3-dependent apoptosis in colon carcinoma cells J. Cancer Res, 2003;24:443–451.
Reddy BS, Kawamori T,Lubet RA, et al. Chemopreventive efficacy of sulindac sulfone against colon cancer depends on time of administration during carcinogenic process J. Cancer Res, 1999;59: 3387–3391.
Samaha HS, Kelloff GT, Steele V, et al. Modulation of apoptosis by sulindac, curcumin, phenylethy 1–3-methylcaffeate and 6-phenylhexyl isothiocyanate: apoptotic index as a biomarker in colon cancer chemoprevention and promotion J.Cancer Res, 1997;57:1301–1305.
Keller JJ, Offerhaus GJ, Drillenburg P, et al. Molecular analysis of sulindac-resistant adenomas in familial adenomatous polyposis J. Clin Cancer Res, 2001; 76: 4000–4007.
Sun BC, Zhao XL, Yan XY, et al. Study of expression influence of sulindac to Fas and Fasl protein in cancerous lesion J. Tianjin Med J, 2003;31:701–704.
Kuwada SK, Scaife CL, Kuang J, et al. Effects of trastuzumab on epidermal growth factor receptor-dependent and independent human colon cancer cells. Int I Cancer, 2004; 109:291–301.
Liu K, McDuffie E, Abrams SI. Exposure of human primary colon carcinoma cells to anti-Fas interactions influences the emergence of pre-existing Fas-resistant metastatic subpopulations. J Immunol, 2003;171:4164–4174.
Eaden J. Review article: the data supporting a role for aminosalicylates in the chemoprevention of colorectal cancer in patients with inflammatory bowel disease. Aliment Pharmacol Ther., 2003;18 Suppl 2:15–21.
Yang W, Bancroft L, Nicholas C, et al.Targeted inactivation of p27kipl is sufficient for large and small intestinal tumorigenesis in the mouse, which can be augmented by a Western-style high-risk diet. Cancer Res, 2003;63:4990–4996.
Kirana C, McIntosh GH, Record IR, et al. Antitumor activity of extract of Zingiber aromaticum and its bioactive sesquiterpenoid zerumbone. Nutr Cancer, 2003;45:218–225.
Stoehlmacher J, Lenz HJ. Cyclooxygenase-2 inhibitors in colorectal cancer. Semin Oncol, 2003;30:10–16.
Author information
Authors and Affiliations
About this article
Cite this article
Sun, B., Zhang, S., Zhao, X. et al. Effects of imbalance of apoptosis and proliferation on large bowel carcinogenesis in mice. Chin. J. Clin. Oncol. 1, 21–26 (2004). https://doi.org/10.1007/BF02739852
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02739852