Abstract
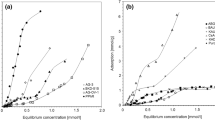
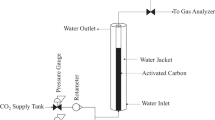
Adsorption isotherms of dichloromethane and 1,1,2-trichloro-1,2,2-trifluoroethane on an activated carbon pellet, Norit B4, were studied. For these chemicals, the Sips equation gave the best fit for the single component adsorption isotherm. The adsorption affinity on activated carbon was greater for dichloromethane than that of 1,1,2-trichloro-1,2,2-trifluoroethane. An experimental and theoretical study was made for the adsorption of dichloromethane and 1,1,2-trichloro-1,2,2-trifluoroethane in a fixed bed. Experimental results were used to examine the effect of operation variables, such as feed concentration, flow rate and bed height. Intraparticle diffusion was able to be explained by a surface diffusion mechanism. An adsorption model based on the linear driving force approximation (LDFA) was found to be applicable to fit the experimental data.
Similar content being viewed by others
References
Chihara, K., Mellot, C. F., Cheetham, A. K., Harms, S., Mangyo, H., Omote, M. and Kamiyama, R., “Molecular Simulation for Adsorption of Chlorinated Hydrocarbon in Zolites,”Korean J. Chem. Eng.,17, 649 (2001).
Cho, S. Y. and Choi, D. K., “Langmuir Parameters for Adsorption of Two Halogenated Chemicals on an Activated Carbon Pellet,”Korean J. Chem. Eng.,13, 409 (1996).
Cicerone, R. J., Stolarski, R. S. and Walters, S., “Stratospheric Ozone Destruction by Man Made Chlorofluoromethanes,”Science,185, 1165 (1974).
Edwards, M. F. and Richardson, J. F., “Gas Dispersion in Parked Beds,”Chem. Eng. Sci.,23, 109 (1968).
Hu, H. B., Yao, S. J., Zhu, Z. Q. and Hur, B. K., “Comparison of the Adsorption Characteristics of Expanded Bed Adsorbent with Conventional Chromatographic Adsorbent,”Korean J. Chem. Eng.,18, 357 (2001).
Hwang, K. S., “Separation of H2/CO2, H2/CO Gas Mixtures by Pressure Swing Adsorption and Fixed-Bed Dynamics,” Ph.D dissertation, KAIST (1994).
Jang, B. H., Lee, S.S., Yeon, T. H. and Tie, T. E., “Effects of Base Metal Promoters in VOC Catalysts for Chlorocarbons and n-Hexane Oxidation,”Korean J. Chem. Eng.,15, 516 (2001).
Lee, D. H. and Moon, H., “Adsorption Equilibrium of Heavy Metals on Natural Zeolites,”Korean J. Chem. Eng.,18, 625 (2001).
Markovska, L., Meshko, V. and Noveski, V., “Adsorption of Basic Dyes in a Fixed Bed Column,”Korean J. Chem. Eng.,18, 190 (2001).
Moon, H. and Tein, C., “Further Work on Multicomponent Adsorption Equilibria Calculations Based on the Ideal Adsorbed Solution Theory,”Ind. Eng. Chem. Res.,26, 2042 (1987).
Na, B. K., Koo, K. K., Eum, H. M., Lee, H. and Song, H. K., “CO2 Recovery from Flue Gas by PSA Process using Activated Carbon,”Korean J. Chem. Eng.,18, 220 (2001).
Ogura, K., Kobayashi, W., Migita, C. T. and Kakum, K., “Complete Photodecomposition of CFC-113, CHC13 and CC14 and Scavenging of Generated Reactive Species,”Environ. Technol.,13, 81 (1992).
Park, I. S., Kwak, C. and Hwang, Y. G., “Frequency Response of Adsorption of a Gas onto Bidisperse Pore-Stuctured Soild with Modulation of Inlet Molar Flow-Rate,”Korean J. Chem. Eng.,18, 330 (2001).
Row, K. H., Choi, D. K. and Lee, Y. Y., “Simulation of the Combined Continuous and Preparative Separation of Three Close-Boiling Components in Gas-Liquid Chromatography,”Korean J. Chem. Eng.,7, 287 (1990).
Ruthven, D. M., “Principles of Adsorption and Adsorption Processes,” Wiley, New York, U.S.A. (1995).
Tien, C., “Adsorption Calculations and Modeling,” Butterworths-Heinemann(1994).
Wang, S. C. and Tien, C., “Further Work on Multicomponent Liquid Phase Adsorption in Fixed Beds,”AIChE J.,28, 565 (1982).
Wakao, N. and Funazkri, T., “Effect of Fluid Dispersion Coefficient on Particle to Fluid Mass Transfer Coefficients in Packed Bed,”J. of Chem. Sci.,33, 1375 (1978).
Wolf, K., “Ozone Depletion and the use of CFCs,”J. Environ. Sci.,32, 41 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S.J., Cho, S.Y. & Kim, T.Y. Adsorption of chlorinated volatile organic compounds in a fixed bed of activated carbon. Korean J. Chem. Eng. 19, 61–67 (2002). https://doi.org/10.1007/BF02706875
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706875