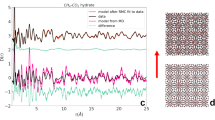
Abstract
Pure methane and nitrogen form structure I and II hydrate, respectively, and therefore the structure type of mixed gas hydrate was found to largely depend on their relative gas composition. In addition to the structural difference of size and shape, each hydrate structure shows different capacity to store the guest molecules. In this study, we investigated phase and structural behaviors according to the composition of methane+nitrogen gas mixture. Three-phase (H-Lw-V) equilibria of solid hydrate, water-rich liquid and vapor phase containing 25.24 mol%, 28.51 mol%, 31.23 mol% and 40.39 mol% of methane were determined at various temperatures (in the range from 273.30 K to 285.05 K) and pressures (from 8.325 MPa to 20.700 MPa). 13C solid-state NMR spectroscopy and powder XRD method were performed to identify the formed structure of hydrate samples. The experimental results showed that gas hydrate of the methane+nitrogen mixture changes its structure from sI to sII between 25.24 mol% and 28.51 mol% of methane concentration. These results of phase behavior and structure identification for the mixed gas hydrates are expected to be very helpful in evaluating the feasibility of exploitation of methane gas from natural gas hydrate and the separation process using gas hydrate as a storage-media
Similar content being viewed by others
References
Jhaveri, J. and Robinson, D. B., “Hydrates in the methane-nitrogen system,”Can. J. Chem. Eng.,43, 75 (1965).
Kim, D. Y., Uhm, T. W. and Lee, H., “Compositional and structural identification of natural gas hydrates collected at site 1249 on ocean drilling program leg 204,”Korean J. Chem. Eng.,22, 569 (2005).
Li, X., Gjertsen, L. H. and Austvik, T., “Thermodynamic inhibitors for hydrate plug melting,”Ann. N.Y.Acad. Sci.,912, 294 (1999).
Mak, T. C. W. and McMullan, R K., “Polyhedral clathrate hydrates. X. structure of the double hydrate of tetrahydrofuran and hydrogen sulde,”J. Chem. Phys.,42(8), 2732 (1965).
McMullan, R. K. and Jeffrey, G A., “Polyhedral clathrate hydrates. IX. structure of ethylene oxide hydrae,”J. Chem. Phys.,42(8), 2725 (1965).
Mei, D. H., Liao, J., Yang, J. T. and Guo, T. M., “Experimental and modeling studies on the hydrate formation of a methane+nitrogen gas mixture in the presence of aqueous electrolyte solutions,”Ind. Eng. Chem. Res.,35(11), 4342 (1996).
Ripmeester, J. A., Tse, T. S., Ratcliffe, C. I. and Powell, B. M., “A new clathrate hydrate structure,”Nature,325, 135 (1987).
Seo, Y T. and Lee, H., “13C NMR analysis and gas uptake measurements of pure and mixed gas hydrates: development of natural gas transport and storage method using gas hydrate,”Korean J. Chem. Eng. 20, 1085 (2003).
Sloan, E. D.,Clathrate hydrates of natural gas, 2nd ed., Dekker, New York, NY (1998).
van Cleeff, A. and Diepen, G M., “Gas hydrates of nitrogen and oxygen,”Rec. Trav. Chem.,79, 582 (1960).
van Klaveren, E. P., Michels, J. J., Schouten, J. S., Klug, D. D. and Tse, J. S., “Stability of doubly occupied N2 clathrate hydrates investigated by molecular dynamics simulations,”J. Chem. Phys.,114, 5745 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, JW., Kim, DY. & Lee, H. Phase behavior and structure transition of the mixed methane and nitrogen hydrates. Korean J. Chem. Eng. 23, 299–302 (2006). https://doi.org/10.1007/BF02705731
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705731