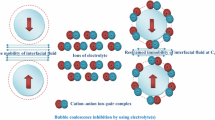
Abstract
Coalescence frequency and coalescence time were measured in electrolyte solutions. Marrucci model was acceptable to predict the coalescence time of one pair bubble in the dilute solution of electrolytes. Transition concentration decreased with increase of bubble forming frequency. This tendency was same as the results in low molecular alcohol solution.
For a bubble column study, the effects of electrolyte on the gas holdup and bubble characteristics were invstigated. The inhibition effect of bubble coalescence of the K2SO4 was slightly higher than that of the KCI at a same ionic strength. In this work, transition concentration was 0.36 kmol/m3, which is larger than the value predicted in pair bubble study.
Similar content being viewed by others
References
Keitel, G. and Onken, V.:Chem. Eng. Commun.,17, 85 (1982).
Walter, J.F. and Blanch, H.W.:Chem. Eng. J,32, B7 (1986).
Drogaris, G. and Weiland, P.:Chem. Eng. Sci.,38(9), 1501 (1983).
Chuang, K.T., Stirling, A.J. and Baker, J.C.:I & EC. Fundam.,23(1), 109 (1984).
Marrucci, G.:Chem. Eng. Sci.,24(6), 975 (1969).
Sagert, N.H. and Quinn, M.J.:Can. J. Chem. Eng.,57, 29 (1979).
Shah, Y.T., Kelkar, S.P., Godbole, S.P. and Deckwer, W.C.:AIChE J.,28(3), 353 (1982).
Andrew, S.P.S.: in International Symposium on Distillation, edited by Rottenburg, D.A., Institution of Chemical Engineering, London (1960).
Kim, J.W. and Lee, W.K.:J. Chem. Eng. Japan,20(5), 448 (1987).
Skelland, A.H.P.: “Diffusional Mass Transfer”, John Wiley & Sons, Inc., New York, U.S.A. (1974).
Walter, J.F.: Ph.D. Dissertation, University of California, Berkeley (1983).
Jin, G.T.: Ph.D. Dissertation, Korea Advanced Institute of Science & Technology (1985).
Kim, J.W. and Lee, W.K.:J. Colloid Interface Sci,123(1), 303 (1988).
Lessard, R.R. and Zieminski, S.A.:Ind. Eng. Chem. Fundam.,10(2), 260 (1971).
Oolman, T.O. and Blanch, H.W.:Chem. Eng. Commun.,43, 237 (1986).
Choi, K.H., Kim, J.W. and Lee, W.K.:Korean J. Chem. Eng.,3(2), 127 (1986).
Himmelblau, D.M.: “Process Analysis by Statistical Methods”, John Wiley, New York, (1970).
Zieminski, S.A. and Whittemore, R.C.:Chem. Eng. Sci.,26, 509 (1971).
Marrucci, G. and Nicodemo, L.:Chem. Eng. Sci.,22, 1257 (1967).
Akita, K. and Yoshida:Ind. Eng. Chem. Process. Des. Develop.,13(1), 84 (1974).
Hikita, H., Asai, S., Tanigawa, K., Segawa, K. and Kitao, M.:Chem. Eng. J.,20, 59 (1980).
Kelkar, B.G., Phulgaonkar, S.R. and Shah, Y.T.:Chem. Eng. J.,27, 125 (1983).
Freedman, W. and Davidson, J.F.:Trans. Inst. Chem. Eng.,47, T251 (1969).
Kim, S.D., Baker, C.G.T. and Bergougnou, M.A.:Can. J. Chem. Eng.,50, 695 (1972).
Hikita, H. and Kikukawa, H.:Chem. Eng. J.,8, 19 (1974).
Bach, H.F. and Philhofer, T.:Germ. Chem. Eng.,1, 270 (1978).
Mersmann, A.:Germ. Chem. Eng.,1, 1 (1978).
Miller, D.N.:AIChE J.,29(2), 312 (1983).
Davidson, L. and Amick, Jr. A.H.:AIChE J.,2, 337 (1956).
Leibson, I., Hocomb, E.G., Cacoso, A.G. and Jacmic, J.J.:AIChE J.,2(3), 296 (1956).
Bhavaraju, S.M., Russell, T.W.F. and Blanch, H.W.:AIChE J.,44, 454 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, J.W., Chang, J.H. & Lee, W.K. Inhibition of bubble coalescence by the electrolytes. Korean J. Chem. Eng. 7, 100–108 (1990). https://doi.org/10.1007/BF02705054
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705054