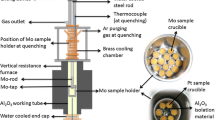
Abstract
The standard free energies of formation of V2C and VC0.73 have been obtained from electromotive force (emf) measurements on the following galvanic cells with BaF2-BaC2 solid solutions as the electrolyte: Ta, Ta2CBaF2-BaC2V, V2 (850 to 1200K) (D) VC0.73, V2C BaF2-BaC2 Cr, Cr23C6 (850 to HOOK) (E) VC0.73, V2C BaF2-BaC2 Mo, Mo2C (890 to 1247 K) (F) Combining the results of this study with previous work1151 and those of Kukarniet al., [19.25] the following equations for ΔGf° of V2C and VC0.73 have been determined: From cell (D), ΔGv2c°(±1263) = -152,824(±9200) + 5.45(±0.27)7 Joule for the reaction 2V + C = V2C. From cell (E), ΔGvc 0.73°(±662) = -96,790.8(±6511.7) + 7.0(±0.3)r Joule/g * atom V From cell (F), ΔGvc 0.73°(±665) = -97,000(±4606) + 6.79(±0.78)J Joule/g * atom V for the reaction V + 0.73C = VC0.73.
Similar content being viewed by others
References
F. A. Shunk:Constitution of Binary Alloys, 2nd Suppl., McGraw-Hill Book Co., New York, NY, 1969, pp. 166–67.
Y.P. Jeannin:Problems of Nonstoichiometry, A. Rabenau, ed., North-Holland Publishing Co., Amsterdam, The Netherlands, 1970, ch. 2, pp. 118–19.
E.K. Storms:The Refractory Carbides, Academic Press, New York, NY, 1967, pp. 48–53.
E.K. Storms and R.J. McNeal:J. Phys. Chem., 1962, vol. 66, pp. 1401–08.
A.D. Mah: U.S. Department of Interior, Bureau of Mines, Report of Inv. No. 6177, Pittsburgh, PA, 1963.
M.A. Guverich:Russ. J. of Inorg. Chem., 1963, vol. 8, pp. 1387–90.
N.M. Volkova and P.V. Gel’d:Tsvetn. Metall., 1963, vol. 5, pp. 89–94.
M.A. Guverich:Russ. J. of Inorg. Chem., 1963, vol. 8, pp. 1390–94.
S. Fujishiro and N.A. Gokcen:J. Electrochem. Soc., 1962, vol. 109, pp. 835–38.
W. Worrell amd J. Chipman:J. Phys. Chem., 1964, vol. 68, pp. 860–66.
A.D. Mah and K.K. Kelly:U.S. Bur. Mines, Rept. Invest., 1961, No. 5858.
J.S. Anderson:The Chemistry of Extended Defects in Non-metallic Solids, L. Eyring and M. O’Keeffe, eds., North-Holland Publishing Co., Amsterdam, The Netherlands, 1970, pp. 1–20.
R. Elliott:Constitution of Binary Alloys, First Supplement, McGraw-Hill Book Co., New York, NY, 1965, p. 704.
P.V. Pillai and M. Sundaresan:Trans Indian Inst. Met., 1975, vol. 28, pp. 319–22.
R.G. Coltters and G.R. Belton:Metall. Trans. B, 1984, vol. 15B, pp. 517–21.
R.G. Coltters and G.R. Belton:Metall. Trans. A, 1983, vol. 14A, pp. 1915–19.
R.G. Coltters and G.R. Belton:Metall. Trans. B, 1980, vol. 11B, pp. 525–29.
O.H. Kriege:The Analysis of Refractory Borides, Carbides Nitrides and Silicides, Los Alamos Scientific Laboratory Report L.A. 2306, Los Alamos, NM, 1959.
M. Atoji:J. Chem. Phys., 1963, vol. 35, pp. 1950–59.
M. Atoji and R.C. Medrud:J. Chem. Phys., 1959, vol. 31, pp. 332–37.
A.D. Kulkarni and W.L. Worrell:Metall. Trans., 1973, vol. 4, pp. 931–33.
V.I. Alekseev and L.A. Schvartman:Dolk. Akad. Nauk SSSR, 1960, vol. 133, p. 1327.
J. Curhbert, R.L. Faircloth, R.H. Flowers, and F.C. Pummery:Proc. Br. Ceram. Soc., 1966, vol. 8, p. 155.
N.M. Volkova and P.V. Gel’d:Izv. Vyssh. Uchebn. Zaved., Tsvetn. Metall., 1965, vol. 3, p. 77.
T. Kireev and R. Karapetyantus:J. Phys. Chem., 1960, vol. 70, p. 68.
A.D. Kulkarni and W.L. Worrell:Metall. Trans., 1972, vol. 3, pp. 2363–70.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coltters, R.G., Nava, Z. High-temperature thermodynamic properties of the vanadium carbides V2C and VC0.73 determined using a galvanic cell technique. Metall Trans B 22, 661–668 (1991). https://doi.org/10.1007/BF02679021
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02679021