Abstract
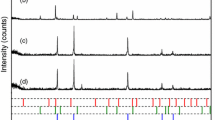
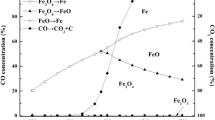
The kinetic features and mechanism of reduction of γ-Fe2O3 in hydrogen in the presence of CaO as a getter for the gaseous reaction products was studied. Data were obtained on the kinetic constants, reaction rate, and evolution of surface area during the reaction. It was established that the getter has a favorable effect on the degree of dispersion of the reaction products.
Similar content being viewed by others
References
V. S. Zenkov, Effect of Preliminary Atomization of the Gas and Additions of Platinum and Palladium on the Reduction of Metal Oxides by Hydrogen, Diss. Kand. Fiz.-Khim Nauk., Kiev (1976).
V. S. Zenkov, “Adsorption processes on getters of the gaseous products of the reduction of metal oxides by hydrogen,” in: Current Achievements in Physical Materials Science [in Russian], Kiev (1995), pp. 42–56.
N. A. Vasyutinskii, Thermodynamics and Kinetics of Metal Reduction Processes [in Russian], Nauka, Moscow (1972), pp. 20–22.
S. T. Rostovtsev, Theory of Metallurgical Processes, Izd-yo N.-T. Lit., Moscow (1956).
V. V. Skorokhod, Yu. M. Solonin, and I. V. Uvarova, Chemical, Diffusion, and Rheological Processes in the Technology of Powder Materials [in Russian], Nauk. Dumka (1990).
B. Del’mon, Kinetics of Heterogenous Reactions [Russian translation], Mir, Moscow (1972).
Additional information
Materials Science Institute, Ukrainian Academy of Sciences, Kiev. Translated from Poroshkovaya Metallurgiya, Nos. 11–12, pp. 21–29, November–December, 1997
Rights and permissions
About this article
Cite this article
Zenkov, V.S., Uvarova, I.V. & Skorokhod, V.V. Influence of getter and gas atmosphere on the reduction kinetics of γ-Fe2O3, and on the phase composition and particle size of the reaction products. Powder Metall Met Ceram 36, 584–590 (1997). https://doi.org/10.1007/BF02676143
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02676143