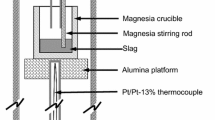
Abstract
Dephosphorization reaction of hot metal by Na2CO3 has been studied experimentally to determine the reaction mechanism and thermodynamics. Most of the experiments were carried out at 1300 °C using Fe-Csat.-Si-P-S alloys. The results indicate that the CO2 gas released from Na2CO3 is important in the dephosphorization reaction as an oxidizer and increasing mass transfer by stirring the slag and metal. As the initial Si content in hot metal is increased, the degree of dephosphorization decreases significantly and the rephosphorization takes place earlier. The primary reason for the rephosphorization is that the activity of PO2.5 increases in the slag because of the evaporation of Na2O from the slag. The loss of Na2O increases the activity coefficient of PO2.5 and decreases the slag volume. At the later stage of Na2CO3 treatment, the reactions reach equilibrium with respect to phosphorus and sulfur, and the oxygen potential,P o2, at the slag-metal interface is determined by the C-CO equilibrium (a c=1 and 1 atm CO). The presence of sulfur in the metal increases the rate of the dephosphorization because of the electrochemical nature of the reaction; sulfur transfer to the slag accepts the electrons from phosphorus transfer.
Similar content being viewed by others
References
S. Anezaki and Y. Shirota:Trans. ISIJ, 1985, vol. 25(7), pp. 652–58.
A. K. Eaton and L. Beck:Geschichte des Eisen, p. 1897.
W. R. Maddocks and E. T. Turkdogan:J. Iron and Steel Inst., 1949, vol. 168, pp. 249–64.
W. J. B. Charter and J. A. Charles:J. Iron and Steel Inst., 1959, vol. 178, pp. 319–28.
W. Oelsen:Arch. Eisenhuettenw., 1965, vol. 36, pp. 861–71.
T. Fuwa:Iron and Steelmaker, June 1981, pp. 18–25.
H. Asai, M. Kanbayashi, R. Kuwahara, and M. Abe:Tetsu-to-Hagané, 1983, vol. 69, pp. 1910–16.
K. Marukawa:Trans. ISIJ, 1980, vol. 20(8), p. 316.
T. Murayama and H. Wada:Proceedings of the Second International Symposium on Metallurgical Slags and Fluxes, TMS-AIME, 1984, pp. 135–52.
J. J. Pak and R. J. Fruehan:Metall. Trans. B, 1986, vol. 17B, pp. 797–804.
A. H. Chan and R. J. Fruehan: Carnegie Mellon University, Pittsburgh, PA, unpublished research, 1987.
T. Moriya and M. Fujii:Trans. ISIJ, 1981, vol. 21, pp. 732–41.
R. Inoue and H. Suito:Trans. ISIJ, 1981, vol. 21(8), pp. 545–53.
F. Tsukihashi, A. Werme, F. Matsumoto, A. Kasahara, M. Yukinobu, T. Hyodo, S. Shiomi, and N. Sano:Proceedings of the Second International Symposium on Metallurgical Slags and Fluxes, TMS-AIME, 1984, pp. 89–106.
R. Inoue and H. Suito:Trans. ISIJ, 1985, vol. 25(2), pp. 118–26.
E. T. Turkdogan and J. Pearson:J. Iron and Steel Inst., 1953, vol. 175, pp. 393–401.
E. T. Turkdogan:Physical Chemistry of High Temperature Technology, Academic Press, Inc., New York, NY, 1980, p. 81.
T. Kawashima, S. Yamaguchi, K. Nagata, and K. S. Goto:J. Japan Inst. Metals, 1984, vol. 48(8), pp. 779–86.
J. Niedringhaus: Ph.D. Thesis, Carnegie Mellon University, Pittsburgh, PA, 1986.
K. S. Goto, S. Yamaguchi, and K. Nagata:Proceedings of the Second International Symposium on Metallurgical Slags and Fluxes, TMS-AIME, 1984, pp. 467–81.
E. T. Turkdogan, G. J. W. Kor, and R. J. Fruehan:Ironmaking Steelmaking, 1980, vol. 7, pp. 268–80.
J. Berg:Canadian Metall. Quart., 1982, vol. 21(2), pp. 121–36.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pak, J.J., Fruehan, R.J. Dynamics of the hot metal dephosphorization with Na2O slags. Metall Trans B 18, 687–693 (1987). https://doi.org/10.1007/BF02672885
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02672885