Abstract
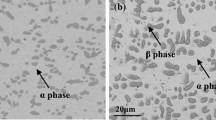
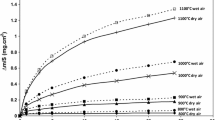
The reaction of alloys with HCI gas is generally more complicated than that of pure metals, and is, typically, a dealloying process. In this work the rates of a model dealloying process, Co(Co-Pt) + 2HC1 → CoCl2(g) + H2 were measured in the temperature range 973 to 1273 K. The only reaction product for the HC1/H2 gas mixtures used in the experiments was gaseous CoCl2. Weight loss during the reaction was continuously monitored with a recording balance. Experimental variables included gas composition, gas flow rate, and temperature for Co-10 at. pct Pt and for pure Co samples. The rate of the reaction was constant with time and very nearly as high for the alloy as for the pure metal, in spite of the fact that Pt was inert for the conditions. A surface instability occurred for the alloy yielding an open porous structure of ever increasing surface area. Transport of CoCl2(g) in the gas boundary layer was important for determining the rate of the chlorination reaction for both the alloy and pure metal.
Similar content being viewed by others
References
R.J. Fruehan:Metall. Trans., 1972, vol. 3, pp. 2585–92.
J.W. Roger:Advances in Corrosion Science and Technology, M. G. Fontana and R. W. Staehle, eds., Plenum Press, vol. 4, p. 245.
J.D. Harrison and C. Wagner:Acta Met., 1959, vol. 7, pp. 722–35.
W.J. Kroll: U.S. Pat. 2,205854, June 1940.
W. A. Henderson:J. Metals, February 1964, vol. 16, pp. 155–64.
Y. Okahara and I. Iwasaki:Trans. SME of AIME, 1970, vol. 247, pp. 73–80.
C. L. Mantell:Tin, Reinhold, New York, NY, 1949, pp. 526–29.
L. Reeve:J. Iron Steel Inst., 1955, vol. 188, pp. 26–40.
E. Martinez:Mining Eng., August 1966, vol. 18, pp. 73–78.
I. Iwasaki, Y. Takahasi, and H. Kahata:Trans. SME-AIME, 1966, vol. 243, pp. 308–20.
R.J. Fruehan and L.J. Martonik:Metall. Trans., 1973, vol. 4, pp. 2789–92.
K. Hauffe and J. Hinrichs:Werkstoffe und Korr., 1970, vol. 21, pp. 954–65.
L. Frommer and M. Polany:Z. Physik. Chem., 1928, vol. 137A, pp. 201–08.
J.D. McKinely:J. Chem. Phys., 1964, vol. 40, pp. 120–25; J.D. McKinely and K. E. Shuler:J. Chem. Phys., 1958, vol. 28, pp. 1207–12; J. N. Smith, Jr. and W. L. Fite:Proc. Int. Symp. Rarified Gas Dyn., 1962, vol. 1, p. 430.
A. Landsberg and F. E. Block: U. S. Dept. Interior, Bureau of Mines, R.I. 6649, 1965.
H. W. Pickering: ”Chlorination of Co-Pt and Fe-Pt Alloys by HC1-H2 Gas Mixtures at 1000 K,” inCorrosion Problems in Energy Conversion and Generation, C.S. Tedmon, Jr., ed., The Electrochm. Soc. Inc., Pennington, NJ, 1974, pp. 151–60.
Y. S. Kim: Ph.D. Dissertation, The Pennsylvania State Univer-sity, 1978.
C. Wagner:J. Electrochem. Soc., 1956, vol. 103, pp. 571–80.
J. D. Harrison and C. Wagner:Acta Met., 1959, vol. 7, pp. 722–35.
B. D. Lichter and C. Wagner:J. Electrochem. Soc, 1960, vol. 107, pp. 168–80.
H. W. Pickering: ”Metallurgical Aspects of Electrolytic Dissolution of Alloys,” inThe Darken Conference, Physical Chemistry in Metallurgy, R.M. Fisher, R. A. Oriani, and E.T. Turkdogan, eds., U.S. Steel Research Laboratory, Monroeville, PA, 1976, pp. 353–73.
H. W. Pickering and Y. S. Kim: ”A Surface Instability During Attack of Co-Pt Alloy by HC1 Gas at Elevated Temperatures and During Electrolytic Dissolution of Cu-Au Alloy at 298 K—Similarities and Differences,” inChemical Metallurgy-A Tribute to Carl Wagner, N. A. Gokcen, ed., AIME, Warrendale, PA, 1981, pp. 455–70.
H.W. Pickering and Y. S. Kim:Corrosion Sci., 1982, vol. 22, pp. 621–35.
M. Hansen:Constitution of Binary Alloys, McGraw-Hill, New York, NY, 1958, pp. 492–94.
R. C. Schoonmaker and R. F. Porter:J. Chem. Phys., 1959, vol. 31, pp. 1586–89.
JANAF Thermochemical Tables Supplement (1975),J. Phys. Chem. Ref. Data, 1975, vol. 4.
H. Wei: M. S. Thesis, The Pennsylvania State University, 1982.
R. Hultgren, T. D. Desai, D. T. Hawkins, M. Gleiser, and K.K. Kelley:Selected Values of the Thermodynamic Properties of Binary Alloys, American Soc. Metals, Metals Park, OH, 1973, p. 681.
E. L. Cussler:Multicomponent Diffusion, Elsevier Scientific Publishing Co., New York, NY, 1976.
A.H.P. Skelland:Mass Transfer, Wiley, New York, NY, 1974.
K. Weil: private communication.
E. R. G. Eckert and R. M. Drake:Heat and Mass Transfer, McGraw-Hill Book Co., New York, NY, 1959.
I. Bann and O. Knacke:Thermochemical Properties of Inorganic Substances, Springer-Verlag, New York, NY, 1973.
Author information
Authors and Affiliations
Additional information
Formerly Research Associate, Department of Materials Science and Engineering, The Pennsylvania State University, University Park, PA 16802.
Rights and permissions
About this article
Cite this article
Kim, Y.S., Pickering, H.W. Kinetics of chlorination of Co and Co-10 At. Pct Pt alloy by reaction with HCI gas. Metall Trans B 13, 349–356 (1982). https://doi.org/10.1007/BF02667750
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02667750