Abstract
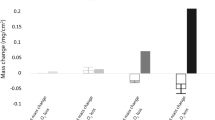
The corrosion rates of nickel in SO2-O2-SO3 atmospheres have been measured to determine corrosion mechanisms under conditions of simultaneous sulfidation-oxidation. The corrosion rates are observed to be independent ofP q2 (0.1 to 0.75 atm) and PSO2 (0.2 to 0.85 atm) but decrease with increasing -PSO3 (0 to 0.17 atm). Although thermodynamic calculations predict the formation of NiSO4, the product scale consists of an inner Ni3S2 layer and an outer two-phase layer of NiO and Ni3S2. The reaction rates are believed to be influenced by both a surface reaction and diffusion through the outer two-phase layer. It is demonstrated that the observed rapid corrosion rates are due to Ni3S2 providing a rapid transport path for nickel diffusion across the outer two-phase layer. When SO3 is present, the corrosion rates decrease due to an extremely irregular distribution of Ni3S2 and a decrease in its fractional area in the two-phase layer. The precise mechanism by which SO3 changes the Ni3S2 distribution is unknown, but it is possible that SO3 reoxidizes part of the chemisorbed sulfur on the scale surface.
Similar content being viewed by others
References
K. L. Lthra and W.L. Worrell:Met. Trans. A, 1978, vol. 9A, pp. 1055–61.
C. B. Alcock, M. G. Hocking, and S. Zador:Corros. Sci., 1969, vol. 9, pp. 111–22.
V. Vasantasree and M. G. Hocking:Corros. Sci., 1976, vol. 16, pp. 261–78.
M. G. Hocking and V. Vasantasree:Corros. Sci., 1976, vol. 16, pp. 279–95.
V. N. Konev, A. M. Borgantsoev, and N. V. Suntsov:Z. Metalloved., 1969, vol. 5, pp. 677–79.
V. N. Konev, V. N. Chebotin, N. V. Suntsov, and L. I. Stavtseva:Z. Metalloved., 1970, vol. 6, pp. 448–50.
T. Rosenqvist:J. Iron Steel Inst., 1954, vol. 176, pp. 37–57.
JANAF Thermochemical Tables, 2nd ed., D. R. Stull and H. Prophet, eds., National Bureau of Standards, Washington, 1971.
V. A. Roiter, N. A. Stukanovskaya, G. P. Korneichuk, N. S. Volikovskaya, and G. I. Golodets:Kinet. Katal., 1960, vol. 1, no. 3, pp. 408–17.
K. Fueki and J. B. Wagner:J. Electrochem. Soc, 1965, vol. 112, pp. 384–88.
E. A. Gulbransen and K. F. Andrew:J. Electrochem. Soc, 1954, vol. 101, pp.128–40.
K. L. Luthra: Doctoral Dissertation, Dept. of Metallurgy and Materials Science, University of Pennsylvania, Philadelphia, August 1976.
H. J. Dall and A. J. Bosnian:Phys. Rev., 1967, vol. 158, pp. 736–47.
H. I. Kaplan and W. L. Worrell:Chemistry of Extended Defects in Non-Metallic Solids, L. Eyring and M. O’Keefe, eds., p. 561, North Holland Publishing Company, 1970.
J. Wagner:Defects and Transport in Oxides, pp. 283–301, Plenum Press, New York, 1973.
K. Kukkola and Wagner:J. Electrochem. Soc, 1957, vol. 104, pp. 379–87.
T. R. Ingraham:Trans. TMS-AIME, 1966, vol. 236, pp. 1064–67.
T. R. Ingraham and P. Marier:Trans. TMS-AIME, 1966, vol. 236, pp. 1067–71.
L. Pauling:The Nature of The Chemical Bond, 3rd ed., p. 329, Cornell University Press, Ithaca, N.Y., 1960.
B. D. Bastow and G. Wood:Oxid. Met., 1975, vol. 9, pp. 473–96.
R. W. G. Wyckoff:Crystal Structure, Interscience Publishers, New York, 1964.
Author information
Authors and Affiliations
Additional information
formerly a Graduate Student, Department of Metallurgy and Materials Science, University of Pennsylvania.
Rights and permissions
About this article
Cite this article
Luthra, K.L., Worrell, W.L. Simultaneous sulfidation-oxidation of nickel at 603°C in SO2-O2-SO3 atmospheres. Metall Trans A 10, 621–631 (1979). https://doi.org/10.1007/BF02658326
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02658326