Abstract
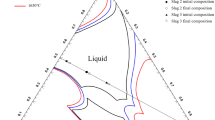
The rate of solution of A12O3 in CaF2 + 30 wt pct A12O3 (at 1518° and 1509°C) and CaF2 + 20 wt pct A12O3 (at 1500°C) liquids has been determined. The operative process is diffusion-controlled, with an interdiffusion coefficient,D for the process varying between 8.5 and 8.1 x 10-5 sq cms - 1 in the CaF2 + 30 wt pct A12O3 slags, and 4.0 × 10-5 sq cms - 1 in the CaF2 + 20 wt pct A12O3 slag. Estimations of the rate at which alumina inclusions would react with these slag during the electroslag processing of steels, indicate that electrode inclusions approaching 100 μ in diam will be dissolved.
Similar content being viewed by others
References
V. G. Levich:Physicochemical Hydrodynamics, Prentice-Hall, Englewood Cliffs, N.J., 1962.
B. Burel: M.A.Sc. Thesis, July 1969, University of British Columbia.
H. Towers and J. Chipman:AIME Trans., 1957, vol. 209, p. 769.
T. G. Pearson: Royal Inst. of Chemistry, Monograph No. 3, p. 43,1955.
P. P. Evseen:Avtomat. Svarka, 1967, vol. 176, p. 42.
V. R. Kammel and H. Winterhager:Erzmetall., 1968, vol. 21, p. 399.
Ling Yang and G. Derge:Physical Chemistry of Process Metallurgy, Pt. I, p. 503, Interscience, 1961.
A Klemm:Molten Salt Chemistry, M. Blander, ed., p. 535, Interscience, 1964.
R. Piontelli:Electrochim. Metall., 1966, vol. 1, p. 191.
R. B. Birdet al: Transport Phenomena, p. 389, John Wiley & Sons, New York, 1962.
S. Joshi and A. Mitchell: University of British Columbia, unpublished research, 1969.
M. M. Klyuev and V. M. Shfitsberg:Steel (USSR), 1969, vol. 2, p. 168.
W. Holzgruber: Ph.D. Thesis, University of Leoban, Austria, 1967.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mitchell, A., Burel, B. The solution rate of alumina in CaF2-Al2 O3 slags. Metall Trans 1, 2253–2256 (1970). https://doi.org/10.1007/BF02643442
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02643442