Abstract
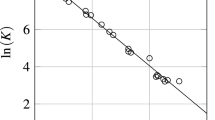
Equations are developed for predicting the activity coefficients of oxygen dissolved in ternary liquid alloys. These are extensions of earlier treatments, and are based on a model in which each oxygen atom is assumed to make four bonds with neighboring metal atoms. It is also postulated that the strong oxygen-metal bonds distort the electronic configuration around the metal atoms bonded to oxygen, and that the quantitative reduction of the strength of bonds made by these atoms with all of the adjacent metal atoms is equivalent to a factor of approximately two. The predictions of the quasichemical equation which is derived agree satisfactorily with the partial molar free energies of oxygen in Ag-Cu-Sn solutions at 1200°C reported in literature. An extension of this treatment to multicomponent solutions is also indicated.
Similar content being viewed by others
References
U. Block and H. P. Stuve:Z. Metallk., 1969, vol. 60, p. 709.
J. L. Meijering:Acta Met., 1957, vol. 5, p. 257.
N. J. Olson and G. W. Topp:Trans. TMS-AIME, 1966, vol. 236, p. 590.
K. T. Jacob and J. H. E. Jeffes:Inst. Mining Met. Trans., London, 1971, vol. 80, p. C32.
K. T. Jacob and C. B. Alcock:Acta Met., 1972, vol. 20, p. 221.
J. D. Bernal:Liquids: Structure, Properties and Solid Interactions, T. J. Hughel, ed., Elsevier, New York, 1965.
C. Diaz, C. R. Mason, and F. D. Richardson:Inst. Mining Met. Trans., London, 1966, vol. 75, p. C183.
C. B. Alcock and T. N. Belford:Trans. Faraday Soc, 1965, vol. 61, p. 443.
J. Carbo-Nover: Ph.D. Thesis, University of London, 1969.
W. A. Fisher and W. Ackermann:Arch. Eisenhüttenw., 1966, vol. 37, p. 697.
K. T. Jacob, S. K. Seshardri, and F. D. Richardson:Inst. Mining Met. Trans. London, 1970, vol. 79, p. C273.
R. J. Fruehan and F. D. Richardson:Trans. TMS-AIME, 1969, vol. 245, p. 1721.
R. Hultgren, R. L. Orr, P. D. Anderson, and Kelley:Selected Values of Thermodynamic Properties of Metals and Alloys, John Wiley and Sons, New York, 1963, and Supplements.
R. Castanet, Y. Claire, M. Gilbert and M. Laffitte:Rev. Int. Hautes Temp. Refract., 1970, vol. 7, p. 51.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jacob, K.T., Alcock, C.B. Prediction of activities of oxygen in dilute quaternary solutions using binary data. Metall Trans 3, 1913–1918 (1972). https://doi.org/10.1007/BF02642578
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02642578