Summary
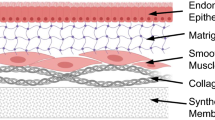
Epithelial cells were isolated from mouse endometrium and cultured on two types of extracellular matrix, namely, rat-tail collagen (type I) gels and basement membrane extract (BME) derived from the Engelbreth-Holm-Swarm murine sarcoma. Cell attachment in serum-free medium during the initial 24 h after seeding was approximately twofold higher on BME compared with collagen type I. Addition of serum to the medium enhanced cell attachment on both matrices. On both collagen and BME, uterine cells grew as smooth-bordered colonies, and within a week of culture the cells became cuboidal to columnar in shape. Electron microscopy revealed the presence of apical microvilli associated with a glycocalyx, junctional complexes, tonofilaments, short strands of undilated endoplasmic reticulum, Golgi complex, and lipid droplets. However, cells on BME showed a higher degree of differentiation as assessed by occasional formation of small patches of basement membranelike structure subjacent to the flattened basal surface and formation of glandlike structures within the matrix. Proliferation of these cells as measured by radioactive thymidine incorporation into DNA was increased threefold by addition of epidermal growth factor (EGF) and insulin to the medium, but was not changed by 17β-estradiol. The expression of progesterone receptors by uterine epithelial cells grown on both matrices was doubled by addition of EGF and estradiol to the medium.
Similar content being viewed by others
References
Burton, K. A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem. J. 62:315–322; 1956.
Carroll, K. M.; Wong, T. T.; Drabik, D. L., et al. Differentiation of rat small intestine epithelial cells by extracellular matrix. Am. J. Physiol. 254:G355-G360; 1988.
Chambard, M.; Gambrion, J.; Mauchamp, J. Influence of collagen gel on the orientation of epithelial cell polarity: follicle formation from isolated thyroid cells and from preformed monolayer. J. Cell Biol. 91:157–166; 1981.
Cooke, P. S.; Uchima, F-D. A.; Fuji, D. K., et al. Restoration of normal morphology and estrogen responsiveness in cultured vaginal and uterine epithelia transplanted with stroma. Proc. Natl. Acad. Sci. USA 83:2109–2113; 1986.
Dawes, C. J. Introduction to biological electron microscopy: theory and techniques. Burlington, VT: Ladd Research; 1988.
Demetris, A. J.; Markus, B. H.; Fung, J. J., et al. Primary culture of human intrahepatic (biliary) epithelial cells. Transplant. Proc. 20:161–163; 1988.
Emerman, J. T.; Pitelka, D. R. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro 13:316–328; 1977.
Errick, J. E.; Kano-Sueoka, T. Growth responsiveness to prolactin and its loss in normal rat mammary cells in culture. Mol. Cell. Endocrinol. 32:255–270; 1983.
Evans, M. I.; McKnight, G. S. Regulation of the ovalbumin gene: effects of insulin, adenosine-3′,5′-monophosphate, and estrogen. Endocrinology 115:368–377; 1984.
Freshney, R. I. Culture of animal cells—a manual of basic technique. New York: Alan R. Liss; 1987.
Glasser, S. R.; Julian, J.; Decker, G. L., et al. Development of morphological and functional polarity in primary cultures of immature rat uterine epithelial cells. J. Cell Biol. 107:2409–2423; 1988.
Hadley, M. A.; Byers, S. W.; Suarez-Quian, C. A., et al. Extracellular matrix regulates Sertoli cell differentiation, testicular cord formation, and germ cell developmentin vitro. J. Cell Biol. 101:1511–1522; 1985.
Haslam, S. Z.; Levely, M. L. Estrogen responsiveness of normal mouse mammary cells in primary culture: association of mammary fibroblasts with estrogenic regulation of progesterone receptors. Endocrinology 116:1835–1844; 1985.
Hsu, Y.-C.In vitro development of whole mouse embryos beyond the implantation stage. In: Daniel, J. C., Jr., ed. Methods in mammalian reproduction. New York: Academic Press; 1978:229–245.
Iguchi, T.; Uchima, F.-D. A.; Ostrander, P. L., et al. Growth of normal mouse vaginal epithelial cells in and on collagen gels. Proc. Natl. Acad. Sci. USA 80:3743–3747; 1983.
Kasis, J. A.; Sakai, D.; Walent, J. H., et al. Primary cultures of estrogen-responsive cells from rat uteri: induction of progesterone receptors and a secreted protein. Endocrinology 114:1558–1566; 1984.
Kleinman, H. K.; Klebe, R. J.; Martin, G. R. Role of collagenous matrices in the adhesion and growth of cells. J. Cell Biol. 88:473–485; 1981.
Kleinman, H. K.; McGarvey, M. L.; Hassell, J. R., et al. Basement membrane complexes with biological activity. Biochemistry 25:312–318; 1986.
Li, M. L.; Aggeler, J.; Farson, D. A., et al. Influence of a reconstituted basement membrane and its components on casein gene expression and secretion in mouse mammary epithelial cells. Proc. Natl. Acad. Sci. USA 84:136–140; 1987.
Mathis, G. A.; Sirica, A. E. Effects of medium and substratum conditions on the rates of DNA synthesis in primary cultures of bile ductular epithelial cells. In Vitro Cell. Dev. Biol. 26:113–118; 1990.
McCormack, S. A.; Glasser, S. R. Differential response of individual uterine cell types from immature rats treated with estradiol. Endocrinology 106:1634–1649; 1980.
McKeehan, W. L.; Barnes, D.; Reid, L., et al. Frontiers in mammalian cell culture. In Vitro Cell. Dev. Biol. 26:9–23; 1990.
Michalopoulos, G.; Pitot, H. C. Primary culture of parenchymal liver cells on collagen mambranes. Exp. Cell Res. 94:70–78; 1975.
Michalopoulos, G.; Slatter, G. L.; Pitot, H. C. Hormonal regulation and the effects of glucose on tyrosine aminotransferase activity in adult rat hepatocytes cultured on floating collagen membranes. Cancer Res. 38:1550–1555; 1978.
Mukku, V. R.; Stancel, G. M. Regulation of epidermal growth factor receptor by estrogen. J. Biol. Chem. 260:9820–9824; 1985.
Quarmby, V. E.; Korach, K. S. The influence of 17b-estradiol on patterns of cell division in the uterus. Endocrinology 114:694–702; 1984.
Richards, J.; Larson, L.; Yang, J., et al. Methods for culturing mammary epithelial cells in a rat tail collagen gel matrix. J. Tissue Cult. Methods 8:31–36; 1983.
Scatchard, G. The attraction of proteins for small molecules and ions. Ann. NY Acad. Sci. 51:660–672; 1949.
Sengupta, J.; Given, R. L.; Carey, J. B., et al. Primary culture of mouse endometrium on floating collagen gels; a potentialin vitro model for implantation. Ann. NY Acad. Sci. 476:75–94; 1986.
Sherman, M. I. Implantation of mouse blastocysts in vitro. In: Daniel, J. C., Jr., ed. Methods in mammalian reproduction. New York: Academic Press; 1978:247–257.
Sumida, C.; Lecerf, F.; Pasqualini, J. R. Control of progesterone receptor in fetal uterine cells in culture: effects of estradiol, progesterone, antiestrogens and growth factors. Endocrinology 122:3–11; 1988.
Tomooka, Y.; DiAugustine, R. P.; McLachlan, J. A. Proliferation of mouse uterine epithelial cellsin vitro. Endocrinology 118:1011–1018; 1986.
Wicha, M. S.; Liotta, L. A.; Kidwell, W. R. Basement membrane collagen requirements for attachment and growth of mammary epithelium. Exp. Cell Res. 124:181–190; 1979.
Yang, J.; Flynn, D.; Larson, L., et al. Growth in primary culture of mouse submandibular epithelial cells embedded in collagen gels. In Vitro 18:435–442; 1982.
Author information
Authors and Affiliations
Additional information
This work was supported in part by a Rockefeller Foundation postdoctoral fellowship (D.G.), and NIh grant 23511.
Rights and permissions
About this article
Cite this article
Ghosh, D., Danielson, K.G., Alston, J.T. et al. Functional differentiation of mouse uterine epithelial cells grown on collagen gels or reconstituted basement membranes. In Vitro Cell Dev Biol – Animal 27, 713–719 (1991). https://doi.org/10.1007/BF02633216
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02633216