Summary
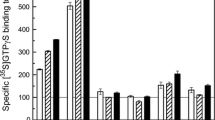
We studied the alterations in the binding of muscarinic cholinergic and adenosine A1 receptors following transient cerebral ischemia in Mongolian gerbils and examined the effects of the novel vinca alkaloid derivative vinconate and pentobarbital against the alterations in the binding of these receptors. Animals were allowed to survive for 5 h and 7 days after 10 min of cerebral ischemia induced by bilateral occlusion of common carotid arteries. [3H]Quinuclidinyl benzilate (QNB) and [3H]cyclohexyladenosine (CHA) were used to label muscarinic cholinergic and adenosine A1 receptors, respectively. The [3H]QNB and [3H]CHA bindings showed no significant alteration in the gerbil brain 5 h after ischemia. However, these bindings in the striatium, the hippocampal CA1 sector, and the hippocampal CA3 sector revealed a significant reduction 7 days after ischemia. The [3H]CHA binding also showed a significant decline in the dentate molecular layer 7 days after ischemia. Intraperitoneal application of vinconate (100 and 300 mg/kg) 10 min and pentobarbital (40 mg/kg) 30 min before ischemia showed a mild reduction in the [3H]CHA binding in the brain 5 h after ischemia. Especially, the reduction was found in the hippocampal CA1 sector and the dentate molecular layer. However, the [3H]QNB binding revealed no significant alteration in the brain 5 h after ischemia. Seven days after ischemia, both drugs prevented a marked reduction in the [3H]CHA binding in the striatium, but not in the hippocampal CA1 sector, the hippocampal CA3 sector, and the dentate molecular layer. By contrast, vinconate and pentobarbital failed to prevent the reduction in the [3H]QNB binding in the striatum. Morphological study indicated that vinconate and pentobarbital ameliorated the neuronal damage to the striatum, but not the hippocampal damage 7 days after ischemia. This histological finding was relatively consistent with the alteration in the [3H]CHA binding. these receptor autoradiographic and histological data suggest that vinconate and pentobarbital can protect the brain from both cellular and functional consequences of ischemia. These findings are of interest in relation to the mechanisms of ischemic brain damage.
Similar content being viewed by others
References
Araki T, Kato H, Kogure K (1989) Selective neuronal vulnerability following transient cerebral ischemia in the gerbil: distribution and time course. Acta Neurol Scand 80:548–553
Araki T, Kato H, Kogure K, Inoue T (1990) Regional neuroprotective effects of pentobarbital on ischemia-induced brain damage. Brain Res Bull 25:861–865
Araki T, Inoue T, Kato H, Kogure K (1990) Neuronal damage and calcium accumulation following transient cerebral ischemia in the rat. Mol Chem Neuropathol 12:203–213
Araki T, Kogure K, Murakami M (1991) Prevention of abnormal calcium accumulation in postischemic gerbil brain by vinconate. Acta Neurol Scand 83:155–160
Araki T, Kato H, Kogure K (1991) Alteration of second messenger systems after transient cerebral ischemia in gerbils: protective effect of pentobarbital and an autoradiographic analysis. Neurosci Lett 130:57–60
Araki T, Kato H, Hara H, Kogure K (1992) Postischemic binding of [3H]phorbol 12,13-dibutyrate and [3H]inostiol-1,4,5-trisphosphate in the gerbil brain: an autoradiographic study. Neuroscience 46:973–980
Bartus RT, Dean III RL, Beer B, Lippa AS (1982) The cholinergic hypothesis of geriatric memory dysfunction. Science 217:408–417
Bonnekoh P, Barbier A, Oschlies U, Hossman HA (1990) Selective vulnerability in the gerbil hippocampus: morphological changes after 5-min ischemia and long survival times. Acta Neuropathol 80:18–25
Cronstein BN, Levin RI, Belanoff J, Weismann G, Hirschorn R (1986) Adenosine: an endogenous inhibitor of neutrophil-mediated injury to endothelial cells. J Clin Invest 78:760–770
Dragunow M, Faull RLM (1988) Neuroprotective effects of adenosine. Trends Pharmacol Sci 9:193–194
Fastbom J, Fredholm BB (1985) Inhibition of [3H]glutamate release from rat hippocampal slices by L-phenylisopropyl-adenosine. Acta Physiol Scand 125:121–123
Foker JE, Einzig S, Wang T (1980) Adenosine metabolism and myocardial preservation: consequence of adenosine catabolism on myocardial high-energy compounds and tissue blood flow. J Thorac Cardiovasc Surg 80:506–516
Fredholm B, Lindgren E, Lindstrom K, Vernet L (1983) The effect of some drugs with purported antianoxic effect on veratridine-induced purine release from isolated rat hypothalamic synaptosomes. Acta Pharmacol Toxicol 53:236–244
Jorgersen MB, Deckert J, Wright DC (1989) Binding of [3H]inositoltrisphosphate and [3H]phorbol 12,13-dibutyrate in rat hippocampus following transient global ischemia: a quantitative autoradiographic study. Neurosci Lett 103:219–224
Kato H, Araki T, Hara H, Kogure K (1991) Sequential changes in muscarinic acetylcholine, adenosine A1 and calcium antagonist binding sites in the gerbil hippocampus following repeated brief ischemia. Brain Res 553:33–38
Lee KL, Tetzlaff W, Kreutzberg GW (1986) Rapid down-regulation of hippocampal adenosine receptor following brief anoxia. Brain Res 380:155–158
Onodera H, Iijima K, Kogure K (1986) Mononucleotide metabolism in the rat brain after transient ischemia. J Neurochem 46:1704–1710
Onodera H, Sato G, Kogure K (1987) Quantitative autoradiographic analysis of muscarinic cholinergic and adenosine A1 binding sites after transient forebrain ischemia in the gerbil. Brain Res 415:309–322
Phillis JW, O'Regan MH (1989) Deoxycoformycin antagonizes ischemia-induced neuronal degeneration. Brain Res Bull 22:537–540
Pulsinelli WA, Brierley JB, Plum F (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11:491–498
Rosdy B, Balazs M, Szporny L (1976) Biochemical effects of ethyl-apovincaminate. Drug Res 26:1923–1926
Rudolphi KA, Keil M, Hinze HJ (1987) Effect of theophylline on ischemically induced hippocampal damage in Mongolian gerbils: a behavioral and histopathological study. J Cereb Blood Flow Metab 7:74–81
Sauer D, Rischke R, Beck T, Roßberg C, Mennel HD, Bielenberg GW, Krieglstein J (1988) Vinpocetine prevents ischemic cell damage in the rat hippocampus. Life Sci 43: 1733–1739
Snyder SH (1985) Adenosine as a neuromodulator. Annu Rev Neurosci 8:103–124
Sternau LL, Lust WD, Ricci AJ, Ratcheson R (1989) Role for γ-aminobutyric acid in selective vulnerability in gerbils, Stroke 20:281–287
Van Calker D, Muller M, Hamprecht B (1979) Adenosine regulates via two different types of receptors, the accumulation of cyclic AMP in cultured brain cells. J Neurochem 33:999–1005
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Araki, T., Kato, H. & Kogure, K. Postischemic alteration of muscarinic acetylcholine and adenosine A1 binding sites in gerbil brain. Res. Exp. Med. 192, 79–88 (1992). https://doi.org/10.1007/BF02576261
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02576261