Abstract
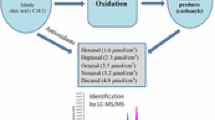
To study the toxicity of low molecular weight (LMW) compounds formed during the autoxidation of oils,14C-labeled primary monomeric compounds (methyl linoleate hydroperoxides) and secondary oxidation products, i.e., polymer and LMW compounds prepared from autoxidized methyl [U-14C]linoleate hydroperoxides (MLHPO) were orally administered to rats, and their radioactive distributions in tissues and organs were compared. The polymeric fraction consisted mainly of dimers of MLHPO. For the LMW fraction, 4-hydroxy-2-nonenal, 8-hydroxy methyl octanoate and 10-formyl methyl-9-decenoate were identified as major constituents by gas chromatography-mass spectrometry (GC-MS) after chemical reduction and derivatization. When LMW compounds were administered to rats,14CO2 expiration and the excreted radioactivity in urine in 12 hr were significantly higher than those from polymer or MLHPO administration. Maximum14CO2 expiration appeared 2–4 hr after the dose of LMW compounds. Radioactivity of the upper part of small intestines six hr after the dose of LMW compounds was higher than the values from administered polymer or MLHPO. The remaining radioactivity in the digestive contents and feces 12 hr after administration of LMW compounds was much lower than the values observed from administered polymer or MLHPO. Among internal organs, the liver contained the highest concentration of radioactivities from polymer, MLHPO and LMW fractions, and an especially higher level of radioactivity was found in liver six hr after the administration of LMW compounds. Six hours after the dose of LMW compounds, a relatively higher level of radioactivity also was detected in kidney, brain, heart and lung. These results show that the LMW compounds from MLHPO autoxidation are more easily absorbed in rat tissues than polymer and MLHPO.
Similar content being viewed by others
References
Nakatsugawa, K., and Kaneda, T. (1983)Yukagaku (J. Jpn. Oil Chem. Soc.) 32, 361–366.
Nakatsugawa, K., and Kaneda, T. (1980)Yukagaku (J. Jpn. Oil Chem. Soc.) 30, 74–77.
Bergan, J.G., and Draper, H.H. (1970)Lipids 5, 976–982.
Findlay, G.M., Draper, H.H., and Bergan, J.G. (1970)Lipids 5, 970–975.
Yoshioka, M., and Kaneda, T. (1972)Yukagaku (J. Jpn. Oil Chem. Soc.) 21, 316–321.
Yoshioka, M., and Kaneda, T. (1974)Yukagaku (J. Jpn. Oil Chem. Soc.) 23, 321–326.
Keppler, J.G., Sparreboom, S., Stroink, J.B.A., and Mikusch, J.D. (1959)J. Am. Oil Chem. Soc. 36, 308–309.
Mizuno, G.R., and Chipault, J.R. (1965)J. Am. Oil Chem. Soc. 42, 839–841.
Johnson, C.B., Pearson, A.M., and Dugan, L.R. Jr. (1970)Lipids 5 958–963.
Freedman, B. (1967)J. Am. Oil Chem. Soc. 44, 113–116.
Miyashita, K., Fujimoto, K., and Kaneda, T. (1982)Agric. Biol. Chem. 46, 751–755.
Neff, W.E., Frankel, E.N., Scholfield, C.R., and Weisleder, D. (1978)Lipids 13, 415–421.
Nakamura, T., Uekihara, N., and Toyomizu, M. (1979)Bull. Jpn. Soc. Sci. Fish. 45, 107–114.
Frankel, E.N. (1980)Prog. Lipid Res. 19, 1–22.
Esterbauer, H., and Schauenstein, E. (1966)Fette Seifen Anstrichm. 68, 7–14.
Bloom, B., Chaikoff, I.L., and Reinhardt, W.O. (1951)Am. J. Physiol. 166, 451–455.
Isselbacher, K.J. (1966)Gastroenterology 50, 78–82.
Kiyasu, J.Y., Bloom, B., and Chaikoff, I.L. (1952)J. Biol. Chem. 199, 415–419.
Nakamura, T., Toyomizu, M., and Nagamoto, T. (1977)Bull. Jpn. Soc. Sci. Fish. 43, 1097–1104.
Nakamura, T., and Toyomizu, M. (1982)Bull. Jpn. Soc. Sci. Fish. 48, 1357–1362.
Schauenstein, E., Esterbauer, H., and Zollner, H. (1977) inAldehydes in Biological Systems (Lagnado, J.R., ed.) pp. 42–51, Pion Limited, London, UK.
Author information
Authors and Affiliations
About this article
Cite this article
Oarada, M., Miyazawa, T. & Kaneda, T. Distribution of14C after oral administration of (U-14C)labeled methyl linoleate hydroperoxides and their secondary oxidation products in rats. Lipids 21, 150–154 (1986). https://doi.org/10.1007/BF02534437
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534437