Abstract
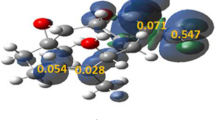
Using the semiempirical quantum-chemical PM3 method, we have studied the formation of a quaternized diazine ring in a series of 4-dimethylaminonaphthalic acid derivatives under Vilsmeier-Haack reaction conditions. We have established that the reaction may occur via a two-step cycloisomerization of the intermediate Vilsmeier adduct, through six-center cyclic transition states.
Similar content being viewed by others
References
B. M. Krasovitskii, L. I. Kormilova, I. G. Ermolenko, et al.,Funct. Mater.,4, No. 2, 280–288 (1997).
D. Barton and W. D. Ollis (eds.),Comprehensive Organic Chemistry [Russian translation], Vol. 4, Khimiya, Moscow (1983).
L. F. Fieser and M. Fieser,Advanced Organic Chemistry [Russian translation], N. S. Vilf'son (ed.), Vol. 2, Khimiya, Moscow (1966).
O. Meth-Cohn and D. L. Taylor,J. Chem. Soc., Chem. Communs., 1463–1464 (1995).
J. J. P. Stewart,J. Comput. Chem.,10, No. 2, 209–232 (1989).
L. D. Patsenker and E. E. Aga,Ukr. Khim. Zh.,64, No. 10, 114–118 (1998).
Additional information
Institute of Single Crystals, National Academy of Sciences of Ukraine, 60 Prospekt Lenina, Khar'kov 310001, Ukraine. Translated from Teoreticheskaya i Éksperimental'naya Khimiya, Vol. 36, No. 4, pp. 201–205, July–August, 2000.
Rights and permissions
About this article
Cite this article
Patsenker, L.D. Theoretical study of the pathway for diazine ring formation in a series of 4-dimethylaminonaphthalic acid derivatives under Vilsmeier-Haack reaction conditions. Theor Exp Chem 36, 183–186 (2000). https://doi.org/10.1007/BF02522748
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02522748