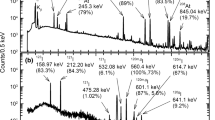
Abstract
A method of radioactivation analysis has been developed for the determination of indium and tin. It is based on substoichiometric extraction of indium diethyldithiocarbamate into carbon tetrachloride from a slightly ammoniacal solution in the presence of potassium cyanide. With this method, indium can be determined via116mIn (T=54 min) and tin via113mIn (T=104 min) which is formed by the reaction112Sn(n, ψ)113Sn. The method has been applied to the determination of indium in metallic zinc and of tin in tin-doped gallium arsenide, and 0.4 ppb of indium was analyzed in a zinc sample.
Similar content being viewed by others
References
J. Růzička, J. Starý,Talanta, 8 (1961) 228; 10 (1963) 287.
N. Suzuki, Proc. 2nd Conf. Radioisotopes (Japan) 1958, p. 151;Sci. Rept. Tohoku Univ., First Ser., 43 (1959) 161.
N. Suzuki, K. Kudo,Anal. Chim. Acta, 32 (1965) 456;K. Kudo, N. Suzuki,Nippon Kagaku Zasshi, 86 (1965) 1050.
J. Růzička, J. Starý, Substoichiometry in Radiochemical Analysis, Pergamon, Oxford, 1968.
J. Starý, J. Růzička,Talanta, 18 (1971) 1.
A. Zeman, J. Starý, J. Růzička,Talanta, 10 (1963) 981.
I. Obrusnik,Talanta, 16 (1969) 563.
S. Bando, T. Imahashi, private communication.
K. Kudo, to be published.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kudo, K., Iso, H. & Shigematsu, T. Subtoichiometric determination of indium and tin by radioactivation analysis. J. Radioanal. Chem. 12, 451–459 (1972). https://doi.org/10.1007/BF02515337
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02515337