Abstract
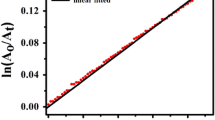
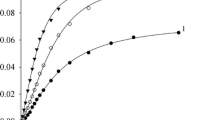
The kinetic behaviour of oximate ions was studied in the transfer of the toluenesulfonyl group in aqueous dimethylsulfoxide (0–95 vol.% DMSO). The solvation effects of the solvent are a factor controlling the nucleophilicity and magnitude of the α-effect of the oximate ions.
Similar content being viewed by others
References
Yu. S. Simanenko, E. A. Karpichev, T. M. Prokop'eva, et al.,Teor. Éksp. Khim.,34, No. 2, 86–90 (1998).
F. Terrier, P. MacCormack, E. Kizilian, et al.,J. Chem. Soc., Perkin Trans. II, No. 1, 153–158 (1991).
R. M. Tarkka and E. Buncel,J. Am. Chem. Soc.,117, No. 5, 1503–1507 (1995).
M. Laloid-Diard, J. -F. Verchore, P. Josselin, et al.,Tetrahedron Lett.,25, No. 12, 1267–1268 (1984).
Yu. S. Simanenko, T. M. Prokop'eva, V. A. Savelova, et al.,Reakts. Sposobn. Org. Soedin.,26, No. 1(93) 30–54 (1989).
Yu. S. Simanenko, T. M. Zubareva, V. A. Savelova, et al.,Teor. Éksp. Khim.,30, No. 6, 319–322 (1994).
A. J. Parker,Advances in Physical Organic Chemistry, No. 5 (1967), pp. 173–185.
W. P. Jencks,Catalysis in Chemistry and Enzymology [Russian translation], Mir, Moscow (1972).
V. A. Savelova, E. A. Karpichev, Yu. S. Simanenko, et al.,Zh. Org. Khim.,32, No. 4, 551–560 (1996).
Additional information
L. M. Litvinenko Institute of Physical Organic and Coal Chemistry, National Academy of Sciences of Ukraine, 70 R. Lyuksemburg ul., Donetsk 340114, Ukraine. Translated from Teoreticheskaya i Éksperimental'naya Khimiya, Vol. 36, No. 1, pp. 30–35, January–February, 2000.
Rights and permissions
About this article
Cite this article
Simanenko, Y.S., Zubareva, T.M., Suprun, I.P. et al. The α-effect of oximate ions and solvent effect in the transfer of the toluenesulfonyl group in aqueous dimethylsulfoxide. Theor Exp Chem 36, 31–36 (2000). https://doi.org/10.1007/BF02511533
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02511533