Abstract
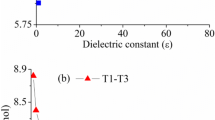
A theoretical study of the mechanism of intramolecular proton transfer reactions in formamide and thioformamide is presented; the focus is on the characterization of the role of water in the reactions. The reaction mechanisms was analyzed with the help of energy profiles in the frame of the reaction force analysis and using the reaction electronic flux to characterize the electronic activity that takes place along the reaction. Bader’s quantum theory of atoms in molecules is used to confirm the reaction mechanism and help elucidate the specific role of water. Results at the DFT/B3LYP 6-311G** level of theory show that water catalyzes the proton transfer reaction lowering the activation energy by a factor of two. The reaction force analysis allowed the characterization of activation energies, indicating that in all four reactions, it is mostly due to structural reordering.







Similar content being viewed by others
References
Bell RP (1980) The tunnel effect in chemistry. Chapman and Hall, New York
Bender ML (1971) Mechanisms of homogeneous catalysis from protons to proteins. Wiley, New York
Boutis T (1992) Proton transfer in hydrogen bonded systems. Plenum, New York
Kim Y, Lim S, Kim HL, Kim Y (1999) J Phys Chem A 103:617
Adamo C, Cossi M, Barone V (1997) J Comput Chem 18:1993
Zielinski TJ, Poirier RA (1984) J Comput Chem 5:466
Wang X-C, Nichols J, Feyereisen M, Gutowski M, Boatz J, Haymet ADJ, Simons J (1991) J Phys Chem 95:10419
Duarte F, Toro-Labbé A (2010) Mol Phys 108:1375
Hargis JC, Vöhringer-Martinez E, Lee Woodcock H, Toro-Labbé A, Schaefer HF III (2011) J Phys Chem A 115:2650
Fu A, Li H, Du D, amd Zhou Z (2003) Chem Phys Lett 382:332
Markova N, Echev VJ (2004) Mol. Struct. (Theochem) 679:195
Fujiwara S, Kambe N (2005) Top Curr Chem 251:87
Vöhringer-Martinez E, Toro-Labbé A (2010) J Comput Chem 31:2642
Bai LL, Yan SH, Ma HQ, Bi SW (2011) Comput Theor Chem 964:218
Toro-Labbé A (1999) J Phys Chem A 103:4398
Gutiérrez-Oliva S, Herrera B, Toro-Labbé A, Chermette H (2005) J Phys Chem A 109:1748
Politzer P, Toro-Labbé A, Gutiérrez-Oliva S, Herrera B, Jaque P, Concha M, Murray J (2005) J Chem Sci 117:467
Rincón E, Jaque P, Toro-Labbé A (2006) J Phys Chem A 110:9478
Labet V, Morell C, Grand A, Toro-Labbé A (2008) J Phys Chem A 112:11487
Herrera B, Toro-Labbé A (2007) J Phys Chem A 111:5921
Echagaray E, Toro-Labbé A (2008) J Phys Chem A 112:11801
Vogt-Geisse S, Toro-Labbé A (2009) J Chem Phys 130:244308
Flores P, Gutiérrez-Oliva S, Silva E, Toro-Labbé A (2010) THEOCHEM 943:121
Duarte F, Toro-Labbé AJ (2011) Chem Phys A 115:3050
Inostroza-Rivera R, Herrera B, Toro-Labbé A (2014) Phys Chem Chem Phys 16:14489
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, New York
Popelier PLA (2000) Atoms in molecules an introduction. Prentice Hall, Upper Saddle River
Brovarets’ OO, Hovorun DM (2013) J Comput Chem 34:2577–2590
Brovarets’ OO, Zhurakivsky RO, Hovorun DM (2014) J Comput Chem 35:451–466
Bonnet ML, Tognetti V (2011) Chem Phys Lett 511:427–433
Parr RG, Yang W (1989) Density functional theory of atoms and molecules. Oxford University Press, New York
Geerlings P, De Proft F, Langenaeker W (2003) Chem Rev 103:1793
Morell C, Tognetti V, Bignon E, Dumont E, Hernandez-Haro N, Herrera B, Grand A, Gutiérrez-Oliva S, Joubert L, Toro-Labbé A (2015) Theor Chem Acc 134:133
Becke A (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr R (1988) Phys Rev B 37:785
Miehlich B, Savin A, Stoll H, Preuss H (1989) Chem Phys Lett 157:200
Vosko S, Wilk L, Nusair M (1980) Can J Phys 58:1200
Fukui K (1981) Acc Chem Res 14:363
Schlegel GW, Scuseria HB, Robb GE, Cheeseman MA, Scalmani JR, Barone G, Mennucci V, Petersson B, Nakatsuji GA, Caricato H, Li M, Hratchian X, Izmaylov HP, Bloino AF, Zheng J, Sonnenberg G, Hada JL, Ehara M, Toyota M, Fukuda K, Hasegawa R, Ishida J, Nakajima M, Honda T, Kitao Y, Nakai O, Vreven H, Montgomery T, Peralta JA Jr, Ogliaro JE, Bearpark F, Heyd M, Brothers JJ, Kudin E, Staroverov KN, Kobayashi VN, Normand R, Raghavachari J, Rendell K, Burant A, Iyengar JC, Tomasi SS, Cossi J, Rega M, Millam N, Klene JM, Knox M, Cross JE, Bakken JB, Adamo V, Jaramillo C, Gomperts J, Stratmann R, Yazyev RE, Austin O, Cammi AJ, Pomelli R, Ochterski C, Martin JW, Morokuma RL, Zakrzewski K, Voth VG, Salvador GA, Dannenberg P, Dapprich JJ, Daniels S, Farkas AD, Foresman Ö, Ortiz JB, Cioslowski JV, Fox J, Frisch DJ, Trucks MJ (2009) Gaussian 09 Revision A.1. Gaussian Inc., Wallingford, CT
Keith TA (2015) Aimall (version 15.05.18). TK Gristmill Software, Overland Park
Giri S, Echegaray E, Ayers PW, Nunez AS, Lund F, Toro-Labbé A (2012) J Phys Chem A 116:10015
Giri S, Inostroza-Rivera R, Herrera B, Nunez AS, Lund F, Toro-Labbé A (2014) J Mol Model 20(9):1–9
Cerón M, Herrera B, Araya P, Gracia F, Toro-Labbé A (2011) J Mol Model 17:1625
Inostroza-Rivera R, Yahia-Ouahmed M, Tognetti V, Joubert L, Herrera B, Toro-Labbé A (2015) Phys Chem Chem Phys 17:17797
Acknowledgments
This work was supported by FONDECYT through project Nos. 1120093, 1100881 and 1130072. The authors acknowledge financial support from ICM through project No. 120082.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published as part of the special collection of articles “CHITEL 2015 - Torino - Italy”.
Rights and permissions
About this article
Cite this article
Guzmán-Angel, D., Inostroza-Rivera, R., Gutiérrez-Oliva, S. et al. Role of water in intramolecular proton transfer reactions of formamide and thioformamide. Theor Chem Acc 135, 37 (2016). https://doi.org/10.1007/s00214-015-1774-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-015-1774-8