Abstract
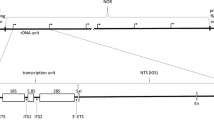
The ribosomal RNA (rRNA) genes of most eukaryotic organisms are arranged in one or more tandem arrays, often termed nucleolar organizer regions. The biological implications of this tandem organization are not known. We have tested the requirement for such a chromosomal organization by directly comparing the transcription and processing of rRNA in isogenic strains ofSaccharomyces cerevisiae that differ only in the organization of their rRNA genes. Strain L-1489 carries theRDN locus, consisting of 100–150 copies of the rRNA genes in a tandem array on chromosome XII. Strain L-1521 has a complete deletion of theRDN array, but carries many copies of a plasmid that includes a single rRNA gene. While this strain grows reasonably well, the average transcriptional activity of the plasmid genes is substantially less than that of the chromosomal copies. However, there is little difference in the processing of the 35S pre-rRNA to the mature 25S:5.8S and 18S products. Thus, neither a chromosomal location nor a tandem repeat of the rRNA genes is important for the assembly and function of the many protein and RNA molecules necessary for the processing of the rRNA transcripts. We investigated the consequence of a dispersed gene arrangement on nucleolar structure. Immunofluorescence microscopy revealed that in strain L-1521 the yeast fibrillarin, Nop1p, rather than being confined to a defined nucleolus at the edge of the nucleus as it is in cells with the normal arrangement of rRNA genes, is spread throughout the nucleus. This observation implies that each plasmid rRNA gene can serve as a nucleolar organizer. Finally, data from pulse-labeling experiments show that the repression of rRNA transcription due to failure of the secretory pathway is independent of whether the rRNA genes are at theRDN locus on chromosome XII or on plasmids. This result suggests that the regulation of rRNA transcription occurs at the level of soluble factors rather than chromatin structure.
Similar content being viewed by others
References
Aris JP, Blobel G (1988) Identification and characterization of a yeast nucleolar protein that is similar to a rat liver nucleolar protein. J Cell Biol 107:17–31
Birnstiel ML, Chipchase M, Speirs J (1971) The ribosomal RNA cistrons. Progr Nucleic Acids Res Mol Biol 11:351–389
Brown DD, Dawid IB (1968) Specific gene amplification in oocytes. Oocyte nuclei contain extrachromosomal replicas of the genes for rRNA. Science 160:272–274
Chernoff YO, Vincent A, Liebman SW (1994) Mutations in eukaryotic 18S ribosomal RNA affect translational fidelity and resistance to aminoglycoside antibiotics. EMBO J 13:906–913
Clark CG, Cross GA (1987) rRNA genes ofNaegleria gruberi are carried exclusively on a 14-kilobase-pair plasmid. Mol Cell Biol 7:3027–3031
Dammann R, Lucchini R, Koller T, Sogo JM (1995) Transcription in the yeast rRNA gene locus: distribution of the active gene copies and chromatin structure of their flanking regulatory, sequences. Mol Cell Biol 15:5294–5303
Elion EA, Warner JR (1986) An RNA polymerase I enhancer inSaccharomyces cerevisiae. Mol Cell Biol 6:2089–2097
Gall JG, Rochaix J-D (1974) The amplified ribosomal DNA of dytiscid beetles. Proc Natl Acad Sci USA 71:1819–1823
Hadjiolov AA (1985) The nucleolus and ribosome biogenesis. Springer, Vienna
Henriquez R, Blobel G, Aris JP (1990) Isolation and sequencing of NOP1. A yeast gene encoding a nucleolar protein homologus to a human autoimmune antigen. J Biol Chem 265: 2209–2215
Johnson SP, Warner JR (1989) Unusual enhancer function in yeast rRNA transcription. Mol Cell Biol 9:4986–4993
Kapler GM (1993) Developmentally regulated processing and replication of theTetrahymena rDNA minichromosome. Curr Opin Genet Dev 3:730–735
Karpen GH, Schaefer JE, Laird CD (1988) ADrosophila rRNA gene located in euchromatin is active in transcription and nucleolus development. Genes Dev 2:1745–1763
Keys DA, Vu L, Steffan JS, Dodd JA, Yamamoto RT, Nogi Y, Nomura M (1994) RRN6 and RRN7 encode subunits of a multiprotein complex essential for the initiation of rDNA transcription by RNA polymerase I inSaccharomyces cerevisiae. Genes Dev 8:2349–2362
Keys DA, Lee BS, Dodd JA, Nguyen TT, Vu L, Fantino E, Burson LM, Nogi Y, Nomura M (1996) Multiprotein transcription factor UAF interacts with the upstream element of the yeast RNA polymerase I promoter and forms a stable preinitiation complex. Genes Dev 10:887–903
Kim RA, Wang JC (1989) A subthreshold level of DNA topoisomerases lead to the excision of yeast rDNA as extrachromosomal rings. Cell 57:975–985
Kiss-László Z, Henry Y, Bachellerie JP, Caizergues-Ferrer M, Kiss T (1996) Site-specific ribose methylation of preribosomal RNA: a novel function for small nucleolar RNAs. Cell 85:1077–1088
Kulkens T, Sande CAFM Van der, Dekker AF, Heerikhuizen H Van, Planta RJ (1992) A system to study transcription by yeast RNA polymerase I within the chromosomal context: functional analysis of the ribosomal DNA enhancer and the RBP1/REB1 binding sites. EMBO J 11:4665–4674
Li BJ, Warner JR (1996) Mutation of the Rab6 homologue ofSaccharomyces cerevisiae, YPT6, inhibits both early Golgi function and ribosome biosynthesis. J Biol Chem 271:16813–16819
Liebman SW, Chernoff YO, Liu R (1995) The accuracy center of a eukaryotic ribosome. In: Matheson AT, Dennis PP, Davies JE, Hill WE (eds) Frontiers in translation. National Research Council of Canada, Ottawa, pp 1141–1149
Liu R, Liebman SW (1996) A translational fidelity mutation in the universally conserved sarcin/ricin domain of 25S yeast ribosomal RNA. RNA 2:254–263
Meier UT (1996) Comparison of the rat nucleolar protein Nopp 140 with its yeast homolog SRP40. J Biol Chem 271: 19376–19384
Melese T, Xue Z (1995) The nucleolus: an organelle formed by the act of building a ribosome. Curr Opin Cell Biol 7:319–324
Mizuta K, Warner JR (1994) Continued functioning of the secretory pathway is essential for ribosome synthesis. Mol Cell Biol 14:2493–2502
Morrow BE, Johnson SP, Warner JR (1993) The rRNA enhancer regulates rRNA transcription inSaccharomyces cerevisiae. Mol Cell Biol 13:1283–1289
Musters W, Venema J, Linden G van der, Heerikhuizen H Van, Klootwijk J, Planta RJ (1989) A system for the analysis of yeast ribosomal DNA mutations. Mol Cell Biol 9:551–559
Nogi Y, Vu L, Nomura M (1991) An approach for isolation of mutants defective in 35S ribosomal RNA synthesis inSaccharomyces cerevisiae. Proc Natl Acad Sci USA 88:7026–7030
Oakes M, Nogi Y, Clark MW, Nomura M (1993) Structural alterations of the nucleolus in mutants ofSaccharomyces cerevisiae defective in RNA polymerase I. Mol Cell Biol 13:2441–2455
Petes TD (1979) Yeast ribosomal DNA genes are located on chromosome XII. Proc Natl Acad Sci USA 76:410–414
Petes TD (1980) Unequal meiotic recombinant within tandem arrays of yeast ribosomal DNA genes. Cell 19:765–774
Ravel-Chapuis P, Nicolas P, Nigon V, Neyret O, Freyssinet G (1985) Extrachromosomal circular nuclear rDNA inEuglena gracilis. Nucleic Acids Res 13:7529–7537
Rose MD, Winston F, Hieter P (1990) Methods in yeast genetics. A laboratory course manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Scheer U, Weisenberger D (1994) The nucleolus. Curr Opin Cell Biol 6:354–359
Schmitt ME, Brown TA, Trumpower BL (1990) A rapid and simple method for preparation of RNA fromSaccharomyces cerevisiae. Nucleic Acids Res 18:3091–3092
Shaw PJ, Jordan EG (1995) The nucleolus. Annu Rev Cell Biol 11:93–121
Thomas BJ, Rothstein R (1989) Elevated recombination rates in transcriptionally active DNA. Cell 56:619–630
Tollervey D, Lehtonen H, Carmo-Fonseca M, Hurt EC (1991) The small nucleolar RNP protein NOP1 (fibrillarin) is required for pre-rRNA processing in yeast. EMBO J 10:573–583
Udem SA, Warner JR (1972) Ribosomal RNA synthesis inSaccharomyces cerevisiae. J Mol Biol 65:227–242
Venema J, Tollervey D (1995) Processing of pre-ribosomal RNA inSaccharomyces cerevisiae. Yeast 11:1629–1650
Warner JR (1990) The nucleolus and ribosome formation. Curr Opin Cell Biol 2:521–527
Warner JR (1991) Labeling of RNA and phosphoproteins inSaccharomyces cerevisiae. Methods Enzymol 194:423–428
Warner JR, Morgan SA, Shulman RW (1976) Kinetics of labeling of the S-adenosylmethionine pool ofSaccharomyces cerevisiae. J Bacteriol 125:887–891
Woolford JL Jr, Warner JR (1991) The ribosome and its synthesis. In: Broach JR, Pringle JR, Jones EW (eds) The molecular and cellular biology of the yeastSaccharomyces Genome dynamics, protein synthesis, and energetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, pp 587–626
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the memory of Asen Hadjiolov, who had great affection for the nucleolus.
Edited by: S.A. Gerbi
Rights and permissions
About this article
Cite this article
Nierras, C.R., Liebman, S.W. & Warner, J.R. DoesSaccharomyces need an organized nucleolus?. Chromosoma 105, 444–451 (1997). https://doi.org/10.1007/BF02510481
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02510481