Abstract
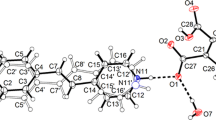
The1H NMR spectra of protonated 1,8-bis(dimethylamino)naphthalene cations (“proton sponges”) containing substituents of various types in positions 2(7) and 4(5) were measured. To estimate the degree of asymmetry of the intramolecular hydrogen bridge in the cations studied, the spin-spin coupling constants of the NH proton with protons of nonequivalent Me2N groups were suggested to be used. Bromination of the “proton sponge” into positions 2 and 7 byN-bromosuccinimide was carried out for the first time.
Similar content being viewed by others
References
V. A. Ozeryanskii and A. F. Pozharskii,Izv. Akad. Nauk, Ser. Khim., 1997, 1501 [Russ. Chem. Bull., 1997,46, 1437 (Engl. Transl.)].
F. Hibbert,J. Chem. Soc., Perkin Trans. 2, 1974, 1862.
A. F. Pozharskii, A. N. Suslov, N. M. Starshikov, L. L. Popova, N. A. Klyuev, and V. A. Adanin,Zh. Org. Khim., 1980,16, 2216 [J. Org. Chem. USSR, 1980,16 (Engl. Transl.)].
H. A. Staab and T. Saupe,Angew. Chem., Int. Ed. Engl., 1988,27, 865.
A. L. Llamas-Saiz, C. Foces-Foces, and J. Elguero,J. Mol. Struct., 1994,328, 297.
Z. Dega-Szafran, B. Nowak-Wydra, and M. Szafran,Magn. Res. Chem., 1993,31, 726.
Z. Malarski, L. Sobczyk, and E. Grech,J. Mol. Struct., 1988,17, 339.
B. Brzezinski, E. Grech, Z. Malarski, and L. Sobczyk,J. Chem. Soc., Faraday Trans., 1990,86, 1777.
B. Brycki, B. Brzezinsky, E. Grech, Z. Malarski, and L. Sobczyk,Magn. Res. Chem., 1991,29, 558.
L. Jaroszewski, B. Lesyng, J. J. Tanner, and J. A. McCammon,Chem. Phys. Lett., 1990,175, 282.
A. F. Pozharskii,Usp. Khim., 1998,67, 3 [Russ. Chem. Rev., 1998,67, 1 (Engl. Transl.)].
J. A. Kanters, A. Schouten, A. J. M. Duisenberg, T. Glowiak, Z. Malarski, L. Sobczyk, and E. Grech,Acta Crystallogr., C, 1991,47, 2148.
J. A. Kanters, E. H. ter Horst, J. Kroon, and E. Grech,Acta Crystallogr., C, 1992,48, 328.
A. F. Pozharskii, V. V. Kuz'menko, G. G. Aleksandrov, and D. V. Dmitrienko,Zh. Org. Khim., 1995,31, 570 [Russ. J. Org. Chem., 1995,31 (Engl. Transl.)].
V. A. Ozeryanskii, A. F. Pozharskii, and N. V. Vistorobskii,Zh. Org. Khim., 1997,33, in press [Russ. J. Org. Chem., 1997,33, in press (Engl. Transl.)].
N. V. Vistorobskii and A. F. Pozharskii,Zh. Org. Khim., 1989,25, 2154 [J. Org. Chem. USSR, 1989,25 (Engl. Transl.)].
S. Toppet, K. Platteborze, and T. Zeegers-Huyskens,J. Chem. Soc., Perkin Trans. 2, 1995, 831.
H. J. G. Yah, M. Soto, and I. Morishima,J. Magn. Reson., 1977,26, 365.
Correlation Analysis in Chemistry: Recent Advances, Eds. N. B. Chapman and J. Shorter, Plenum Press, New York-London, 1978.
V. Balasubramaniyan,Chem. Rev., 1966,66, 567.
The Chemistry of the Amino Group, Ed. S. Patai, Interscience Publ., London-New York-Sydney, 1968, 161.
B. I. Ionin, B. A. Ershov, and A. I. Kol'tsov, inYaMR-spektroskopiya v organicheskoi khimii [NMR Spectroscopy in Organic Chemistry], Khimiya, Leningrad, 1983, 56 (in Russian).
H. A. Staab, M. A. Zirnstein, and C. Krieger,Angew. Chem., Int. Ed. Engl., 1989,28, 86.
A. F. Pozharskii, N. L. Chikina, N. V. Vistorobskii, and V. A. Ozeryanskii,Zh. Org. Khim., 1997,33, in press [Russ. J. Org. Chem., 1997,33, in press (Engl. Transl.)].
E. M. Arnett, inProgress in Physical Organic Chemistry, Eds. S. G. Cohen, A. Streitwieser, and R. W. Taft, Interscience Publ., New York-London, 1963.
N. V. Vistorobskii and A. F. Pozharskii,Izv. Akad. Nauk, Ser. Khim., 1996, 2297 [Russ. Chem. Bull., 1996,45, 2181 (Engl. Transl.)].
Author information
Authors and Affiliations
Additional information
For Part 22, see Ref. 1.
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 68–75, January, 1998.
Rights and permissions
About this article
Cite this article
Pozharskii, A.F., Ozeryanskii, V.A. peri-Naphthylenediamines. Russ Chem Bull 47, 66–73 (1998). https://doi.org/10.1007/BF02495511
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02495511