Abstract
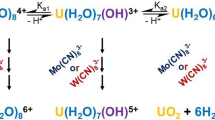
Photooxidation of water by the uranyl ion was studied. Solutions of uranyl in 0.01–4.0M H2SO4, HClO4, or 0.1–1.0M Na2SO4 and NaClO4 containing “lacunary” heteropolytungstate (HPT) K10P2W17O61 or K8SiW11O39 were irradiated with a nitrogen laser, a mercury or xenon lamp, or visible light. Spectrophotometric analysis showed that the irradiation results in the accumulation of UIV. Simultaneously the formation of H2O2 proceeds. The quantum yield Φ of the reaction increases as the concentration of the acid or salt increases. For aerated solutions of 1M H2SO4 or 1M HClO4, irradiation by light with λ=337.1 Φ is close to (1.5–2)·10−3. The irradiation of solutions with pH −4 for many days leads to an almost quantitative transformation of UO2 2+ into UIV. When the irradiation was carried out in the absence of HPA, UIV was not detected, although hydrogen peroxide was observed in the solution.
Similar content being viewed by others
References
H. D. Burrows and T. J. Kemp,Chem. Soc. Rev., 1974,3, 139.
A. L. Buchachenko and I. V. Khudyakov,Usp. Khim., 1991,60, 1105 [Russ. Chem. Rev., 1991,60 (Engl. Transl.)].
H. A. Schwarz and R. W. Dodson,J. Phys. Chem., 1984,88, 3643.
U. K. Klänning, K. Schested, and J. Holeman,J. Phys. Chem., 1985,89, 760.
M. Moriyasu, Y. Yokoyama, and S. Ikeda,J. Inorg. Nucl. Chem., 1977,39, 2211.
V. N. Parmon, inFotokataliticheskoe preobrazovanie solnechnoi energii [Photocatalytic Conversion of Solar Energy], Ch. 2, Nauka, Novosibirsk, 1985, 6 (in Russian).
M. D. Marcantonatos,Inorg. Chim. Acta, 1977,24, L37.
L. A. Khamidullina, S. V. Lotnik, and V. P. Kazakov,Izv. Akad. Nauk, Ser. Khim., 1994, 751 [Russ. Chem. Bull., 1994,43, 704 (Engl. Transl.)].
M. P. Souchay,Polyanions et Polycations, Gauthier, Paris, 1963.
C. A. Parker,Photoluminescence of Solutions, Elsevier Publishing Company, Amsterdam, 1968.
L. Bion, Ph. Moisy, and C. Madic,Radiochimica Acta, 1995,69, 251.
J. T. Kiwi and M. Daniels,J. Inorg. Nucl. Chem., 1978,40, 576.
E. A. Erin, A. A. Baranov, A. Yu. Volkov, V. M. Chistyakov, and G. A. Timofeev,Radiokhimiya, 1995,37, 115 [Radiochemistry, 1995,37 (Engl. Transl.)].
J.-M. Adnet,Extraction Selective des Actinides des Effluents de Haute Activite, These, Centre d'Etudes Nucleaire de Fontenay-aux-Roses, 1992.
J. C. Sullivan, S. Gordon, D. Cohen, W. Mulac, and K. H. Schmidt,J. Phys. Chem., 1976,80, 1684.
The Chemistry of the Actinide Elements, Vol.1, 2nd Eds. J. J. Katz, G. T. Seaborg, and L. R. Morss, Chapman and Hall, London-New York, 1986.
Y. Mao and A. Bakac,Inorg. Chem., 1996,35, 3925.
L. P. Maslov, L. V. Sirotinkina, and A. G. Rykov,Radiokhimiya, 1985,27, 732 [Radiochemistry, 1985,27 (Engl. Transl.)].
W.-D. Wang, A. Bakac, and J. H. Espenson,Inorg. Chem., 1995,34, 6034.
D. Meisel, Y. A. Ilan, and G. Czapski,J. Phys. Chem., 1974,78, 2330.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya. No. 2, pp. 282–287, February, 2000.
Rights and permissions
About this article
Cite this article
Yusov, A.B., Shilov, V.P. Reduction of the photoexcited uranyl ion by water. Russ Chem Bull 49, 285–290 (2000). https://doi.org/10.1007/BF02494673
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02494673