Abstract
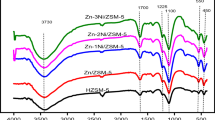
The acidity and coke deposits of HZSM-5 and NiHZSM-5 catalysts were studied in the aromatization using NH3-TPD, IR and TPD techniques. The results showed that both strong acid and B acid sites decreased quickly at the initial stage of reaction for the two catalysts. The coking and reaction rates also dropped quickly at this stage. Yet it was discovered, that adding Ni into HZSM-5 led to weakening B acid sites and strengthening L acid ones. At the same time, the results revealed that metallic Ni decreases the coking rates and slows down the activity decline rates.
Similar content being viewed by others
References
Zhu Guangzhong, Liu Wenming, Chen Zhaoping:Acta Petrol. Sin., (Petroleum Processing Section),12, 103 (1996).
Zhu Guangzhong:Acta Petrol. Sin., (Petroleum Processing Section)13, 100 (1997).
Jiang Yi, Liang Juan Zhao Sugin:J. Catal., (Cuihua Xuebao),15, 463 (1994).
Zhuo Runsheng, Cheng Changrui, Tan Changyu:J. Chem. Techn.,23, 180 (1995).
Zhu Guangzhong:J. Jiangxi Univ.,17, 53 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guangzhong, Z., Ping, Z. A study of acidity and coke deposits on NiHZSM-5 catalyst in aromatization. React Kinet Catal Lett 66, 231–236 (1999). https://doi.org/10.1007/BF02475795
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02475795