Summary
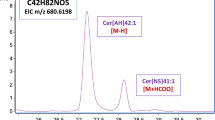
This paper describes the development of a chromatographic system for analysis of commercial ceramides structurally similar to those found in the stratum corneum. The ceramides used in this study contain different amine based (phytosphingosine, sphingosine and dihydrosphingosine) and fatty acids of different chain lengths and with different functional groups (hydroxylated and unsaturated). Non-aqueous reversed-phase (NARP) liquid chromatography with evaporative light-scattering detection (ELSD) were the techniques chosen in accordance with the nature of the ceramides. The eluent strength and the potential selectivity of different organic solvents were investigated. On a C18-bonded silica, the most promising chromatographic conditions employed a gradient from ACN-THF, 95∶5, to ACN-THF-PrOH, 35∶5∶60, in 15 min with a constant concentration of TEA (10 mM) and a stoichiometric amount of formic acid.
Similar content being viewed by others
References
D. T. Downing, J. Lipid Res.33, 301 (1992).
K. J. Robson, M. E. Stewart, S. Michelsen, N. D. Lazo, D. T. Downing, J. Lipid Res.35, 2060 (1994).
K.-A. Karlsson, I. Pascher, J. Lipid Res.12, 466 (1971).
B. C. Melnik, M. D. J. Hollmann, E. Erler, B. Verhoeven, G. Plewig, The Society for Invest. Dermatol.2, 231 (1989).
S. Motta, M. Monti, S. Sesana, R. Caputo, S. Carelli, R. Ghidoni, Biochimica Biophysica Acta1182, 147 (1993).
D. g. Paige, N. Morse-Fisher, J. I. Harper, Br. J. Dermatol.131, 23 (1994).
J. T. Bernett, N. K. Meredith, J. R. Akins, W. H. Hannon, J. Liq. Chromatogr. Rel. Technol.8, 1573 (1985).
W. W. Christie, J. Lipid Res.26, 507 (1985).
C. A. Demopoulos, M. Kyrili, S. Antonopoulou, N. K. Andrikopoulos, J. Liq. Chromatogr. Rel. Technol.19, 771 (1996).
T. Gildenast, J. Lasch, Biochimica Biophsica Acta1346, 69 (1996).
M. Previati, L. Bertolaso, M. Tramarin, V. Bertagnolo, S. Capitani, Anal. Biochem.233, 108 (1996).
M. Iwamori, C. Costello, H. W. Moser, J. Lipid Res.20, 86 (1979).
U. H. Do, P. T. Pei, Lipids16, 855 (1981).
S. Heron, A. Tchapla, Analusis21, 269 (1993).
J. L. Robinson, R. Macrae, J. Chromatogr.303, 386 (1984).
H. Lambers, W. van der Wilden, F. Farin, W. Keuning, Parfums Cosmetiques122, 51 (1995).
S. Heron, A. Tchapla, J. Chromatogr.556, 219 (1991).
G. Thevenon-Emeric, A. Tchapla, M. Martin, J. Chromatogr.550, 267 (1991).
A. Tchapla, S. Heron, E. Lesellier, H. Colin, J. Chromatogr. A656, 81 (1993).
P. J. Schoenmakers, H. A. H. Billiet, L. De Galan, J. Chromatogr.185, 179 (1979).
L. R. Snyder, J. W. Dolan, J. R. Gant, J. Chromatogr.23, 3 (1979).
N. Chen, Y. Zhang, P. Lu, J. Chromatogr.603, 35 (1992).
P. Chaminade, A. Baillet, D. Ferrier, J. Chromatogr. A672, 67 (1994).
R. Kaiser, in Gas-chromatographie, Geest und Portig, Leipzig, 1960, p. 33; and inP.J. Schoenmakers, Optimization of Chromatographic Selectivity, Elsevier, Amsterdam, Nederlands, 1986, vol. 35, p. 119.
K. Gaudin, P. Chaminade, A. Baillet, D. Ferrier, J. Bleton, S. Goursaud, A. Tchapla, J. Liq. Chromatogr.22, 379 (1999).
M. Dreux, M. Lafosse, L. Morin-Allory, LC-GC9, 148 (1996).
G. Guiochon, A. Moysan, C. Holley, J. Liq. Chromatogr.11, 2547 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gaudin, K., Chaminade, P., Ferrier, D. et al. Analysis of commercial ceramides by non-aqueous reversed-phase liquid chromatography with evaporative light-scattering detection. Chromatographia 49, 241–248 (1999). https://doi.org/10.1007/BF02467550
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02467550