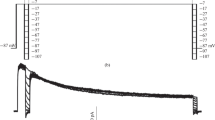
Abstract
Tetrodotoxin-sensitive and tetrodotoxin-resistant sodium currents were studied in rat dorsal root ganglion neurons using a patch-clamp method. The type of hydrogen ion buffer used was found to affect the kinetics of the inactivation process. Tris ions irreversibly bound to the inactivation gating apparatus, accelerating the decay in the trailing front of the ion current. The characteristics of this process were clearly nonlinear in this buffer. In HEPES buffer, the inactivation dynamics were slowed for both types of channel studied. Unlike results obtained with tris buffer, there were no sharp changes in the characteristics as compared to those obtained immediately after puncture of the cell membrane. The advantages of using HEPES buffer for studies of the inactivation gating processes of sodium channels are discussed.
Similar content being viewed by others
References
N. S. Veselovskii, P. G. Kostyuk, and A. Ya. Tsyndrenko, “Discrimination of ion currents responsible for generating action potentials in the somatic membranes of neurons in newborn rats,” Dokl. Akad. Nauk SSSR,249, No. 6, 1466–1469 (1979).
Yu. I. Zil'berter and E. N. Timin, “The process of inactivation of rapid sodium currents through membranes of single cardiac cells,” Byull. Éksp. Biol. Med.,9, No. 10, 5–7 (1982).
B. V. Krylov, “Modeling of membrane adaptation at nodes of Ranvier as a result of sodium channel inactivation,” Neirofiziologiya,15, No. 5, 535–539 (1983).
A. V. Lonskii, Z. I. Krutetskaya, and N. G. Roshchina, “The effect of niflumic acid on components of the inactivation of sodium channels in the membranes of nodes of Ranvier,” Fiziol. Zh. SSSR,75, No. 3, 327–333 (1989).
A. Marti and E. Neer, Whole-Cell Recording in Conditions of Dense Contacts. Recording of Single Channels [Russian translation], Moscow (1987).
G. N. Mozhaeva, A. P. Naumov, and E. D. Nosyreva, “The kinetics of the decay in the sodium current on repolarization of the axon membrane in normal conditions and in the presence of scorpion toxin,” Neirofiziologiya,12, No. 5, 541–549 (1980).
E. M. Peganov, “On the kinetics of the process of inactivation of sodium channels in the frog node of Ranvier,” Byull. Éksp. Biol. Med.,76, No. 11, 5–9 (1973).
J. Somien, Encoding of Sensory Information in the Mammalian Nervous System [Russian translation], Moscow (1975).
G. N. Akoev, N. P. Alekseev, and B. V. Krylov, Mechanoreceptors. Their Functional Orgnaization, Berlin (1988).
B. Bromm, J. R. Schwartz, and G. Ochs, “A quantitative analysis of combined potential and current clamp experiments of the single myelinated nerve fibre ofRana esculenta,” J. Theoret. Neurobiol.,1, No. 1, 120–133 (1981).
G. Chen, A. E. Cole, A. B. MacDermott, et al., “The influence of skeletal muscle on the electrical excitability of dorsal root ganglion neurons in culture,” Neuroscience,7, No. 8, 2412–2422 (1987).
S. Y. Chiu, “Inactivation of sodium channels: second order kinetics in myelinated nerve,” J. Physiol.,273, No. 3, 573–596 (1977).
A. A. Elliott and J. R. Elliott, “Characterization of TTX-sensitive and TTX-resistant sodium currents in small cells from adult rat dorsal root ganglia,” J. Physiol.,463, 39–56 (1993).
O. P. Hamill, A. Marty, E. Neher, et al., “Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches,” Pflug. Arch.,391, No. 1, 85–100 (1981).
J. W. Hanrahan and J. A. Tabcharani, “Inhibition of an outwardly rectifying anion channel by HEPES and related buffers,” J. Membr. Biol.,116, No. 2, 65–77 (1990).
A. L. Hodgkin and A. F. Huxley, “The dual effect of membrane potential on sodium conductance in the giant axon of Loligo,” J. Physiol.,116, No. 4, 497–506 (1952).
K.-D. Kniffki, D. Siemen, and W. Vogel, “Development of sodium permeability inactivation in nodal membranes,” J. Physiol.,313, 37–48 (1981).
P. G. Kostyuk, N. S. Veselovsky, and A. Y. Tsyndrenko, “Ionic currents in the somatic membrane of rat dorsal root ganglion neurons. 1. Sodium currents,” Neuroscience,6, No. 12, 2423–2430 (1981).
B. V. Krylov, S. A. Kirov, S. A. Podzorova, and G. N. Akoev, “Additional state of the sodium channel inactivation system: probable mechanism for spike frequency adaptation in sensory neurons,” Primary Sensory Neuron,1, No. 1, 3–35 (1995).
P. Niemann, J. Schmidtmayer, and W. Ulbricht, “Chloramine-T effect on sodium conductance of neuroblastoma cells as studied by whole-cell clamp and single-channel analysis,” Pflüg. Arch.,418, No. 1-2, 129–136 (1991).
W. Nonner, “Relations between the inactivation of sodium channels and the immobilization of gating charge in frog myelinated nerve,” J. Physiol.,299, 573–603 (1979).
G. Ochs, B. Bromm, and J. R. Schwarz, “A three-state model for inactivation of sodium permeability,” Biochem. Biophys. Acta,645, No. 2, 243–252 (1981).
Y. V. Osipchuk and E. N. Timin, “Electrical measurement on perfused cells,” in: Intracellular Perfusion Neuron of Excitable Cells, London (1984).
I. H. Peacock, P. G. Nelson, M. W. Goldstone, “Electrophysiological study of cultured neurons dissociated from spinal cord and dorsal root ganglia of fetal mice,” Dev. Biol.,30, No. 1, 137–152 (1973).
M. L. Roy and T. Narahashi, “Differential properties of tetrodotoxin-sensitive and tetrodotoxin-resistant sodium channels in rat dorsal root ganglion neurons,” J. Neuroscience,12, No. 6, 2104–2111 (1992).
J. Schmidtmayer, “Voltage and temperature dependence of normal and chemically modified inactivation of sodium channels. Quantitative description by a cyclic three-state model,” Pflüg. Arch.,414, No. 3, 273–281 (1989).
J. R. Schwartz, B. Bromm, R. P. Spielmann, and J. L. F. Weytjens, “Development of Na inactivation in motor and sensory myelinated nerve fibres ofRana esculenta,” Pflüg. Arch.,398, No. 2, 126–129 (1983).
R. P. Spielmann, J. R. Schwartz, and B. Bromm, “Oscillating repolarization in action potentials of frog sensory myelinated nerve fibers,” Neurosci. Lett.,36, No. 1, 49–53 (1983).
Additional information
Laboratory of Reception Physiology, I. P. Pavlov Institute of Physiology, Russian Academy of Sciences, 6 Makarov Bank, 199034 St. Petersburg. Translated from Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 82, No. 7, pp. 1–10, July, 1996.
Rights and permissions
About this article
Cite this article
Krylov, B.V., Podzorova, S.A. & Vilin, Y.Y. Inactivation kinetics of sensory neuron sodium channels depend on the type of hydrogen ion buffer. Neurosci Behav Physiol 28, 65–71 (1998). https://doi.org/10.1007/BF02461914
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02461914