Summary
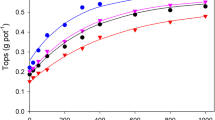
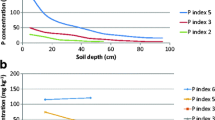
A simple model is described, and experimentally tested, for predicting the rate of dissolution of rock phosphates in soil, including the effect of solubilization by plant roots. The sensitivity of the model to its input parameters is assessed and it is seen that plants may significantly increase their P uptake by acid secretion. The model provides a rational basis for the selection of P solubilizing crops and inter-crops.
Similar content being viewed by others
References
Kirk G J D and Nye P H 1985 The dissolution and dispersion of dicalcium phosphate dihydrate in soils. I. A predictive model for a planar source. J. Soil Sci. 36, 445–459.
Kirk G J D and Nye P H 1985 The dissolution and dispersion of dicalcium dihydrate in soils. II. Experimental evaluation of the model. J. Soil Sci. 36, 461–468.
Kirk G J D and Nye P H 1986 The dissolution and dispersion of dicalcium dihydrate in soils. III. A predictive model for regularly distributed particles. J. Soil Sci. 37, 511–524.
Kirk G J D and Nye P H 1986 The dissolution and dispersion of dicalcium phosphate dihydrate in soils. IV. Experimental evaluation of the model for particles. J. Soil Sci. 37, 525–528.
Kirk G J D and Nye P H 1986 A simple model for predicting the rates of dissolution of sparingly soluble calcium phosphates in soil. I. The basic model. J. Soil Sci. 37, 529–540.
Kirk G J D and Nye P H 1986 A simple model for predicting the rates of dissolution of sparingly soluble calcium phosphates in soil. II. Applications of the model. J. Soil Sci. 37, 541–554.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nye, P.H., Kirk, G.J.D. The mechanism of rock phosphate solubilization in the rhizosphere. Plant Soil 100, 127–134 (1987). https://doi.org/10.1007/BF02370936
Issue Date:
DOI: https://doi.org/10.1007/BF02370936